May 26, 2022
I. introduction
We live in a vast universe. A universe that undoubtedly extends far beyond even the visible limits we can perceive, given the finite speed at which light can propagate throughout the universe. A visible universe that contains more than 100 billion galaxies, each of which may contain 100 billion or more stars, as does the Milky Way. From a scientific viewpoint, then, it is highly improbable that Earth is the only site in that vast universe on which life exists in some form.
Life begins with the assembly of biomolecules that are capable of storing genetic information and undergoing replication, with DNA and RNA as the particular examples that have fueled the evolution of species on Earth. The initial assembly of those biomolecules relies on chemical elements that are ubiquitous throughout the universe and energy available from nearby stars. Those building blocks interact through universal laws of chemistry and physics. We do not yet understand completely the detailed mechanisms by which those biomolecules originated on Earth, but we believe it can happen via random interactions of the constituents, given enough time and a suitable environment to support those interactions. Earth has a favorable environment, but it is hard to believe that it is a unique environment in the vast universe.
And yet, within the history of human experience – keeping in mind that human civilizations have existed on Earth for less than one millionth of the age of the universe – we have not yet seen any convincing evidence of contact with living species from other planets. This lack of evidence led the great physicist Enrico Fermi once to muse “But where is everybody?” Up until 30 years ago, we had no direct evidence even that other planets existed outside our own solar system. But the search for such exoplanets has accelerated rapidly and very successfully during the current century, while searches for possible signals of extra-terrestrial intelligence have also expanded. In this series of posts we survey recent progress and further advances anticipated for the coming decades on both types of searches — the focus of large teams of modern explorers. Parts I and II are devoted to efforts to identify other Earth-like planets within the Milky Way galaxy that may potentially provide environments suitable for the development of life.
II. conditions needed for life to develop
What we know of life comes from our only source of evidence, observations of life and experiments on Earth. The following brief description of the conditions that favor the development of life repeats some of the points and text appearing in the book Signatures of the Artist, authored by one of us (SV).
Assembling the building blocks:
The basic building blocks of biomolecules are organic compounds, such as amino acids or nucleotides, which are composed from hydrogen, carbon, oxygen and nitrogen atoms, and sometimes heavier elements. The neutrons and protons that assemble to form the nuclei of those atoms were themselves formed during the first microseconds of the universe’s existence, and they combined to form the lightest nuclei during the first several minutes. Hydrogen atoms were formed in abundance throughout the expanding universe during the so-called recombination epoch – some 380,000 years after the Big Bang – when the universe had cooled sufficiently that electrons electrically attracted to attach themselves to protons were unlikely to be subsequently unseated (see our post on Young Earth Creationism). Carbon, nitrogen and oxygen are produced in the fusion processes contributing to energy production in stars, as are heavier elements up to iron in some stars. Elements heavier than iron appear to be produced primarily in cataclysmic cosmic events, including supernova explosions and neutron star mergers. All of these processes and all of these elements occur throughout the universe.
There is well-established experimental evidence that organic compounds would have been synthesized from these abundant elements under the volatile conditions that should have characterized Earth’s atmosphere during its first billion years. (The age of the Earth is well determined to be about 4.5 billion years.) In the seminal experiment, carried out by Stanley Miller and Harold Urey at the University of Chicago in 1953, a mixture of water vapor (H2O), methane (CH4), ammonia (NH3), and hydrogen (H2) gases was exposed to electrical discharge sparks intended to simulate lightning interacting with Earth’s early atmosphere (see Fig. II.1). After two weeks of continuous operation, Miller and Urey found evidence for the synthesis of 11 different amino acids – an estimate later revised by subsequent reanalyses of the same Miller-Urey samples to more than the 20 amino acids that occur naturally in proteins of life forms on Earth.

Subsequent experiments have demonstrated that the production of amino acids and diverse other organic compounds would have been a feature as well of other atmospheric compositions, including ones containing carbon dioxide, nitrogen, hydrogen sulfide, and sulfur dioxide, molecules that would have been injected into the atmosphere by major volcanic eruptions. Experiments and simulations have also demonstrated that ultraviolet light from the Sun and geothermal heating would have provided effective alternatives to lightning as the energy sources needed to drive the early organic compound formation. Furthermore, amino acid content similar to that generated in the Miller-Urey experiment has been found in samples of meteorites that have struck Earth. So the simple organic compounds needed for life exist beyond Earth, as well.
It is still a very long way from the synthesis of relatively simple organic molecules, such as amino acids and nucleotides (so-called monomers) to the formation of living cells. Monomers must combine with one another to form the long polymer chains characteristic of biological processes: for example, typical proteins comprise several hundred amino acid monomers. The crucial step leading to biologically viable systems and to biological evolution is the synthesis of complex macromolecules capable of storing genetic information and of being faithfully replicated in molecular processes. In the living systems we observe today, the relevant macromolecules are DNA, RNA and enzyme proteins that catalyze the chemical reactions needed to replicate DNA. But there is general agreement that the chemical basis for the earliest life on Earth probably was based on simpler and shorter macromolecules.
Both experiments and theoretical considerations of RNA chemistry suggest that it is possible for assemblies of roughly 50 ribonucleotides (like those that are the building blocks of RNA and DNA) to act as ribozymes that are capable of catalyzing their own self-replication as information-storing systems. Such assemblies could have served as the earliest biomolecules. In 1996 Ferris, Hill, Liu and Orgel demonstrated the natural stepwise synthesis of polymers of about that length, in a period of 1—2 weeks (later reduced to 1—2 days), on mineral surfaces embedded in water containing a mixture of the relevant monomers. Specifically, the experiment by Ferris and his collaborators utilized naturally occurring clay minerals formed by the weathering of volcanic ash, which tend to adsorb organic compounds such as nucleotides, thereby enhancing their local concentration, and to catalyze reactions among them. The adsorption on mineral surfaces tended to overcome the hydrolysis – the breaking of bonds between monomers by the addition of water molecules – that had doomed previous attempts to synthesize long polymers in aqueous solutions.
Early ribozymes that may have formed over time in such a stepwise natural assembly from ambient monomers need not have been capable at first of highly efficient or accurate self-replication. Indeed, errors in the replication would have led to mutations, which occasionally produced ribozymes that were capable of faster and more accurate self-replication, and which therefore came to dominate subsequent samples. In a classic 1993 experiment, David Bartel and (2009 Nobel Laureate) Jack Szostak demonstrated that catalytic activity among nucleic acid polymers (i.e., candidate RNA molecules) increased rapidly over time by such mutation and natural chemical selection processes.
The central role of water:
Such experiments demonstrate at least a plausible path for life to have begun from initially lifeless building blocks that may have accumulated in water with mineral surfaces on the early Earth. They help to flesh out Charles Darwin’s original speculation that life on Earth might have begun through chemistry “in some warm little pond, with all sorts of ammonia and phosphoric salts, light, heat, electricity, etc. present, so that a protein compound was chemically formed ready to undergo still more complex changes.” The experiments emphasize the central role accorded in searches for other Earths to the existence of liquid water on the surface of a planet.
Why is liquid water viewed as a requirement for life to begin and flourish? The most important physical characteristic of water for biology is its ability to dissolve and transport almost any substance, thereby providing an environment that supports the encounters of different chemical compounds and the biochemical reactions needed for an organism to develop and evolve. These reactions are at the heart of complex molecule formation and of reproductive, cell division, and metabolic processes. They are catalyzed by enzymes that can attain the requisite three-dimensional shape in a water environment, as needed to speed up the biochemical reactions.
Furthermore, water readily absorbs radiation from nearby stars, leading to molecular vibrations, but its high heat capacity limits the resulting temperature increase. So, liquid water provides a moderating influence on climate. Its existence in the liquid state extends over a sufficiently large temperature and pressure range that day-to-night and seasonal variations have little effect on its overall abundance. But at the same time, its ability to evaporate and form clouds, and then to recondense into rain or snow, and for snow and ice to melt into the liquid state – all within not too large a temperature range – is critical to the transport of water from oceans to land and back again. Water’s unusual property of expanding and becoming less dense when it freezes keeps ice on the ocean’s surface, where it can subsequently melt, rather than sinking and gradually filling up underwater spaces, where life also thrives. Its high surface tension facilitates droplet formation, enabling plants to extract water-borne nutrients from soil.
This collection of properties makes water optimally suited to the development of life forms. But admittedly, our conceptions of the requirements for life are extrapolated from “Earth-bound” experience. Some planetary scientists have speculated that perhaps extraterrestrial life could develop – not without considerable difficulty – in liquid ammonia or methane, in the absence of water and its unique set of physical properties. But for now, most research looking for potential other sites for life in our galaxy focuses on the search for rocky planets, comparable in size to Earth and capable of supporting liquid oceans on the surface.
The Habitable Zone:
The most basic requirement for a planet to host liquid water on its surface is that it fall within a “Goldilocks” or Habitable Zone of distances from the star the planet orbits, where its surface temperature is neither too cold nor too hot, but just right for water to maintain the liquid state. As illustrated by the green band in Fig. II.2, the range of suitable distances depends strongly on the surface temperature of the star. The distances in Fig. II.2 are measured in astronomical units (AU), where the mean distance of Earth from the Sun is defined to be 1 AU.
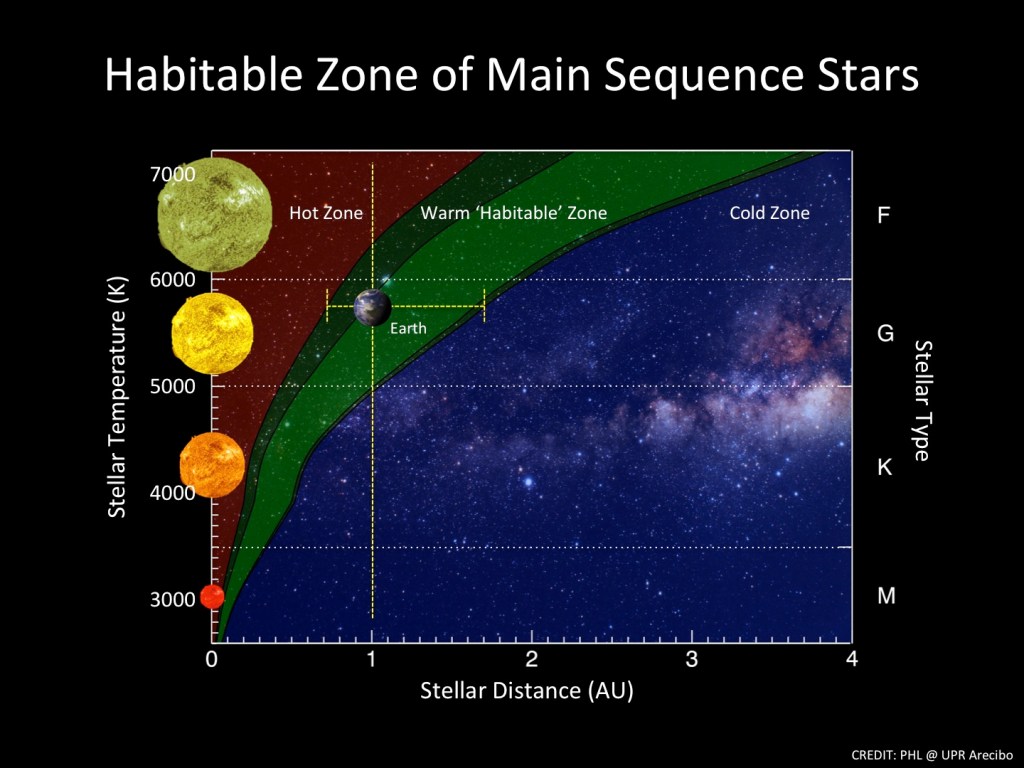
The classification of stars indicated on the right-hand side of Fig. II.2 refers to the Hertzsprung-Russell diagram of Fig. II.3, in which the so-called Main Sequence of stars is indicated by the tight correlation of surface temperature with stellar brightness. As indicated by slanting lines and labels in Fig II.3, those stellar features are also correlated with the size and predicted lifetime of the star. The larger and hotter the star, the more rapidly it exhausts its hydrogen supply in stellar fusion processes, and so, the shorter its lifetime.

Most indications are that life on Earth began about a billion years after the Sun began to burn hydrogen. So stars above the M, K, G and F classes indicated at the bottom of Fig. II.3, with predicted lifetimes of a billion years or less, are not considered promising candidates to host habitable planets. The majority of stars in Earth’s general vicinity within the Milky Way galaxy are, in fact, small and relatively dim M-class stars, a number of them labeled in Fig. II.3. As we will see in section IV, a number of planets in tight orbits have already been identified within the habitable zone of several of these M-class stars. But as we will also see, there are some questions about the ability of these M dwarf stars to support life in their planetary systems.
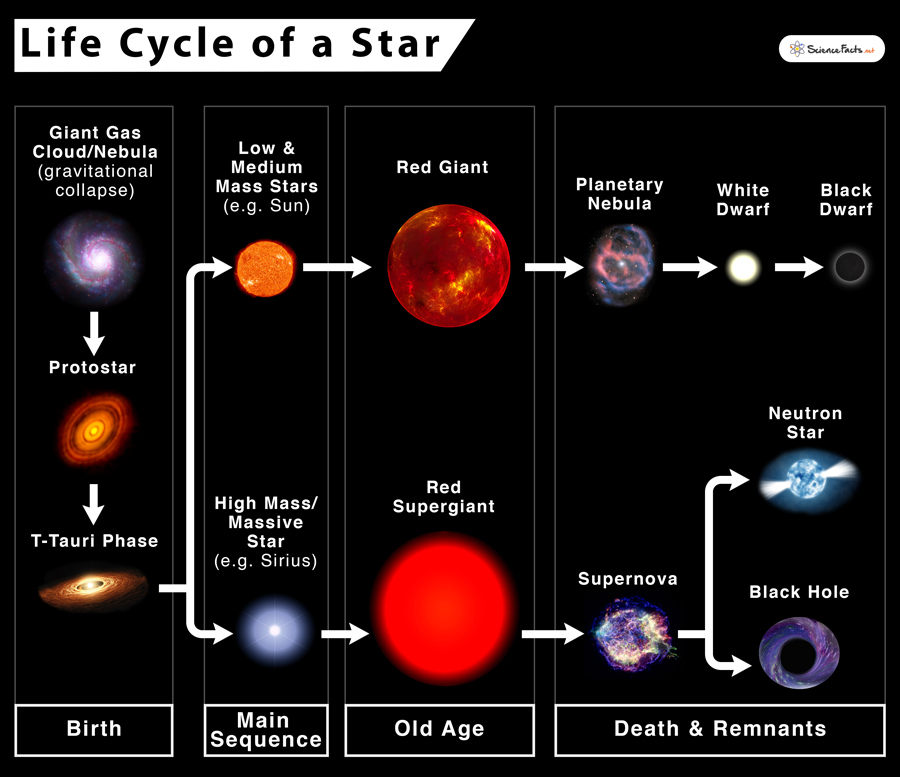
The stars off the main sequence curve in Fig. II.3 represent late stages in the stellar life cycles illustrated in Fig. II.4, and are not likely to host habitable planets. Low- and medium-mass main sequence stars will ultimately evolve into red giants when they have exhausted the hydrogen fuel in their cores. They subsequently convert energy from gravitational collapse into burning of hydrogen in the surrounding stellar atmosphere. During that phase the stars swell up to 100 or 1000 times the size of our Sun, and their surface burns cooler, thus appearing red. They can still generate energy in their cores by the fusion of helium nuclei, but when the helium is exhausted they shed their outer layers, turning into a planetary nebula and, eventually, collapsing into a white dwarf star. More massive main sequence stars get hot enough in their cores that they can continue producing energy by fusing heavier nuclei – producing elements all the way up to iron – even after their helium is exhausted, and they swell to red supergiants of truly spectacular size.
The importance of Earth’s atmosphere:
Not all stars that fall within the habitable range of distances from their stars can actually support life. The environment on Earth illustrates a number of other relevant features. Surface oceans can remain bound by gravity to even a spinning planet, but surface water can evaporate under irradiation from the nearby star. Thus, atmospheric pressure is essential to maintain surface liquid water over long time periods. The atmosphere then participates in the water cycle, recondensing evaporated water to provide precipitation that can deliver nutrients to life on land. An appropriate atmosphere can also help to shield the surface from some deep ultraviolet radiation from a star like the Sun and other cosmic radiation, which might be harmful to living organisms. A magnetosphere, such as Earth has, is also useful in this regard, diverting charged particle radiation headed toward the planet.
The atmosphere also contains gases needed for living organisms. We are, of course, most used to thinking about oxygen needed for breathing of humans and other animals, but life began on Earth when there were few, if any, oxygen molecules in its atmosphere. In the earliest stages of planet formation, planets are surrounded by hot gases containing the most abundant molecules in the universe, namely hydrogen and helium. But in Earth’s case, those light molecules moved fast enough in the hot proto-atmosphere to escape Earth’s gravity. The atmosphere was then replenished with the help of frequent volcanic eruptions as Earth’s crust was forming and tectonic plates were shifting. The volcanoes released water vapor, carbon dioxide, ammonia and sulfates, which dominated the early atmosphere. Absorption of radiation from the Sun eventually broke up most of the ammonia into nitrogen molecules, which remain, and hydrogen molecules, which have mostly escaped the atmosphere.
Sulfates and carbon dioxide, dissolved into the oceans from that early atmosphere, provided fuel for the earliest living organisms, single-celled bacteria and archaea in the oceans. The oceans eventually filled with cyanobacteria that got their energy by photosynthesis: absorbing sunlight and carbon dioxide in the water, to produce carbohydrates and release oxygen. It was the output from those cyanobacteria that led to the Great Oxygenation Event in Earth’s atmosphere, which recent geochemical research has pinned down to a period as short as one to ten million years in duration, occurring 2.33 billion years ago. That rapid buildup of oxygen in the atmosphere launched the proterozoic geologic era, during which multicellular organisms formed, leaving us the first obvious fossil evidence of life on Earth, and leading eventually to the Cambrian explosion, when fossils of most major animal classes began to appear. Oxygen molecules (O2) in the atmosphere also led to the production of ozone (O3) in the stratosphere, which is produced by, but also absorbs much of the most harmful UV radiation from the Sun, shielding the emergence of complex forms of life on the surface.
What we learn, then, from the reconstructed history of Earth is that oxygen and ozone are produced in the atmosphere by living organisms and fuel further co-dependent evolution of life forms and the atmosphere. The presence of oxygen and ozone in a planet’s atmosphere is not essential for living organisms to begin, but can be considered as one component of a biosignature that photosynthesis is occurring on the planet and complex life forms can be supported. Photosynthesis of life forms on a planet is also critical for absorbing carbon dioxide from the atmosphere. As should be clear from our present concerns about global warming of Earth, too much CO2 in the atmosphere leads to too much absorption of infrared thermal radiation from the planet, causing a runaway greenhouse effect, and eventually warming the planet too much for it to support life. This is, for example, the situation on Venus, whose thick, toxic CO2-dominated atmosphere makes it the hottest planet in our solar system. Earth’s atmosphere also benefits from being relatively thin, as seen in Fig. II.5, allowing all but deep UV radiation from the Sun to penetrate to the surface.

A thin atmosphere could potentially get frozen on a planet that strays too far from its star in a highly eccentric (i.e., elongated) orbit, or on one orbiting so near its star that it becomes tidally locked, as the Moon is to Earth, with a permanent dark side that remains cold even if the planet itself resides in the habitable zone. So, Earth’s nearly circular orbit around the Sun and its daily rotation about its axis also help to maintain liquid water on its surface.
The above considerations determine what we look for in a search for other Earths: rocky planets, of size and density roughly comparable to Earth’s, orbiting small to medium-mass main sequence stars in low-eccentricity orbits within the habitable zone; thin atmospheres in which absorption of starlight indicates a chemical composition reflecting volcanic activity, water vapor from surface oceans, possible photosynthesis and other signs of life. In the current stage of the ongoing research, to be described in section IV, surveys are identifying candidates whose atmospheres will subsequently be studied in detail with new and upcoming space and ground-based telescopes.
III. techniques to detect exoplanets
It is extremely challenging to image exoplanets directly because their light is normally dwarfed by that of the star they orbit, and their angular separation from the star, as viewed from Earth, is quite small. For calibration, a planet that orbits a star 40 light years away from Earth, in an Earth-like orbit of radius 1 AU (the distance from Earth to our Sun), has an angular separation of 0.08 arc seconds as viewed from Earth, right around the excellent resolution limit of the recently launched James Webb Space Telescope. If the star is similar to the Sun and the planet to the Earth, then starlight reflected from the planet has an intensity within the visible light spectrum only about 1 part in 10 billion of that from the star itself. It might at first seem advantageous to image the exoplanets in their longer-wavelength deep infrared thermal emission spectrum, rather than in reflected starlight, since there the light intensity ratio improves by typically four orders of magnitude; the cooler planet has an emission spectrum peaked at much larger wavelengths than the hotter star. However, a telescope’s ability to resolve the planet from the star worsens in the deep infrared because the fundamental diffraction limit on resolution increases in direct proportion to the wavelength of the light received.
Thus, the vast majority of exoplanets that have been discovered to date were detected via their indirect influence on the light from the star. In fact, the first definitive discovery of exoplanets in a quite unexpected planetary system came in 1992, when Wolszczan and Frail detected tiny periodic perturbations to the otherwise extremely regular radio wave pulses received in the Arecibo radio telescope every 6.2 milliseconds from a pulsar 2300 light years away from Earth. They attributed the perturbations to the orbits of two planets orbiting the pulsar: one with mass 3.4 times that of Earth, orbiting at a distance of 0.36 AU with an orbital period of 66.6 days; the second with 2.8 Earth masses, at 0.47 AU and orbit period of 98.2 days.
Pulsars are rapidly rotating, highly magnetized neutron stars. They emit narrow beams of electromagnetic radiation from their magnetic poles, and that radiation can be detected on Earth whenever that beam sweeps past Earth like a rotating lighthouse beam. In this case, the orbital motion of the exoplanets caused recoil of the pulsar that slightly altered the otherwise steady period with which the pulsar beam swept by Earth. Planets surrounding pulsars are unexpected because neutron stars are the remnants of supernova explosions (see Fig. II.4) that tend to destroy any planets surrounding the progenitor star. And since neutron stars are dead stars, hosting no nuclear fusion, the discovered planets are not habitable.
Subsequent early discoveries were made by detecting the periodic motion of stars toward or away from Earth caused by orbiting exoplanets. In everyday language, we say that Earth revolves around the Sun. But more precisely, the Earth and the Sun exert equal and opposite gravitational forces on each other and, in the absence of other planets, both would revolve with the same period about their common center of mass. Since the Sun is so much more massive than the Earth, that center of mass is quite close to the center of the Sun, so the Sun’s motion is over much smaller distances than Earth’s and at an orbital velocity opposite in direction to and smaller than that of Earth by the ratio of the Earth mass to the Sun mass. (Of course, the Sun is responding to gravitational pulls from all the planets in the solar system.) If a distant star has a comparable planet-induced wobble that leads to periodic changes in the star’s radial velocity toward or away from Earth, then that velocity change results in a tiny Doppler shift up and down in the wavelength of light received on Earth from the star (see Fig. III.1). That Doppler shift is the analog of the change in sound wave frequency you hear from the engine hum of a race car approaching you versus receding from you.
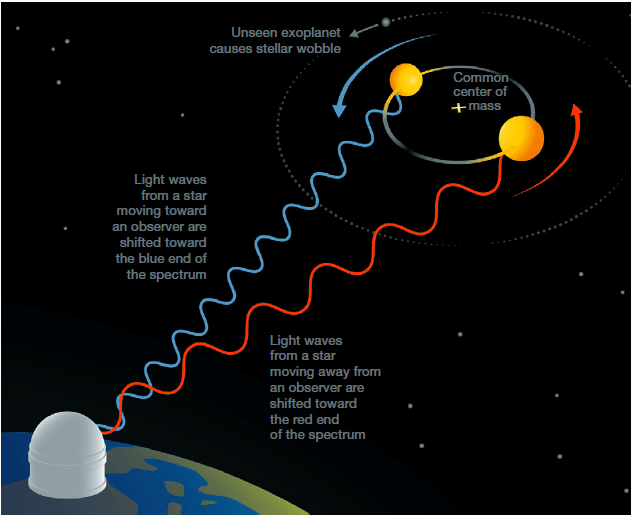
For an Earth-like planet orbiting a Sun-like star that radial velocity can be tiny with respect to the speed at which light propagates, and hence the wavelength shift can be too small to detect. Earth’s speed around its orbit is about 30 kilometers per second, and the correlated speed of the Sun is only 9 centimeters per second, or 3 parts per 10 billion of the speed of light. But the star’s radial velocity is proportional to the mass of the orbiting planet. (It depends weakly on the planet’s orbit period, being inversely proportional to the cube root of the period.) So this method works best for giant planets. Indeed, the first discovery of a Jupiter-like gas giant exoplanet around a Sun-like star (51 Pegasi) in the Milky Way was made using this technique in 1995 by Michel Mayor and Didier Queloz, using the Haute-Provence Observatory in southern France. Since this seminal discovery launched an era of exponential increase in the number of exoplanet discoveries (see Fig. III.2), Mayor and Queloz were awarded one half of the 2019 Nobel Prize in Physics.

From the period observed in the Doppler wavelength oscillations, the radial velocity method can directly determine the orbital period of the planet. If the star’s mass is known by other methods, the magnitude of the Doppler shift can then determine the planet’s mass. But this technique does not tell you the size or density of the exoplanet. The workhorse of exoplanet discoveries during this century has been a different technique, looking for dips in the starlight received by a telescope when an orbiting planet, in a favorably oriented orbit plane, passes between the star and the telescope. This transit method is illustrated in Fig. III.3, using real data obtained by NASA’s Spitzer Space Telescope. These data herald the discovery of an exoplanet in such a tight orbit around the star LHS 3844 that the planet’s “year” lasts only 11.1 hours. The Spitzer viewed the star and planet in the near infrared portion of the spectrum edge-on with the planet’s orbit. The data in Fig. III.3 span one complete orbit (though the data are averaged over many orbits).

The transit of the planet across our line of sight to the star leads in Fig. III.3 to a 0.5% dip in the infrared light intensity, a dip that recurs every 11.1 hours. The magnitude of that dip tells us the size of the planet, because it reveals the fraction of the star’s area that the planet eclipses. If the same exoplanet can also be studied by the radial velocity method, one can then determine the planet’s density from its size and mass.
Over about half of the orbit, between labeled points 6 and 12 in Fig. III.3, the space telescope picks up starlight reflected from the planet’s surface in addition to that directly from the star itself. That reflected light leads to a gentle rise in total light intensity over most of that half of the orbit, until the planet passes behind the star, where it is eclipsed. The size of the resulting secondary dip from that planet eclipse tells us the reflected brightness, or planetshine, of the transiting planet. If the planetshine spectrum in light wavelength can be measured with sufficient precision, we can learn about the nature of the planet’s surface and atmosphere, as we will explore further below.
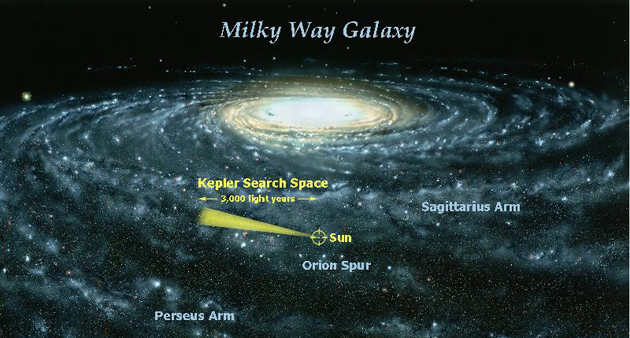
The search for exoplanets in transit received an enormous boost with the 2009 launch of the Kepler Space Telescope. Kepler and its follow-up incarnation K2 continuously monitored the light output from hundreds of thousands of stars – looking for exoplanet transit dips – within a narrow region of the Milky Way galaxy (see Fig. III.4) over the years from 2009 to 2018. And it discovered thousands of new exoplanets covering a variety of types, including a number comparable in size to Earth and residing within the habitable zone of their local stars. We will review some statistics about the Kepler discoveries in section IV, but a flavor for the excitement they produced can be seen in a video on the NASA website. The frequency of new discoveries suggests that a sizable fraction of stars in the galaxy host planets and planetary systems. The Kepler mission was subsequently extended with the Transiting Exoplanet Search Satellite (TESS), launched in 2018 to survey the entire sky over a period of two years, focusing on some 200,000 of the brightest stars in the Milky Way.
Yet another method for detecting exoplanets, used to date by a number of ground-based observatories, exploits the gravitational influence of both a star and its orbiting planet on the light from a more distant star further from Earth along the same line of sight. The gravitational field of massive objects (whether they shine themselves or not) bends light in a somewhat similar way to the refraction of light in earthbound lenses. For example, a foreground star can act as a gravitational lens to magnify light from a background star or galaxy that passes behind it. A properly positioned telescope will detect a spike in light intensity as the background star passes behind the foreground star. A planet orbiting the foreground star will add lensing mass and gravity when it passes the line of sight, causing a secondary, smaller spike in the light from the background star. This technique for detecting exoplanets and determining their mass is known as gravitational microlensing.
Microlensing will be exploited in exoplanet searches by the future Nancy Grace Roman Space Telescope. As suggested by its former name as the Wide Field Infrared Survey Telescope (WFIRST), the Roman, though similar in size to the Hubble Space Telescope, will have 100 times wider view of the sky in infrared radiation. The Roman will also feature a state-of-the-art optical system that can serve as a coronagraph, “a system of masks, prisms, detectors and even self-flexing mirrors built to block out the glare from distant stars — and reveal the planets in orbit around them” by direct imaging. Direct imaging of exoplanets has been used sparingly to date by ground-based telescopes, and only to view very large and relatively bright planets in orbits with tens of AU radius around their stars. An example is shown in Fig. III.5. In contrast, the Roman coronagraph will be “capable of seeing planets that are almost a billion times fainter than their star.”

An alternative to coronagraph optics, intended to provide another new technology capable of revolutionizing direct imaging of exoplanets, is the starshade currently under development at Caltech’s Jet Propulsion Laboratory. This is an origami-like structure that can be carried in a compact mode by a future space telescope. When the telescope focuses on a particular star, it can launch the starshade ahead of it on the sight line to the star, where the starshade will spring outward and unfold into a sunflower shape illustrated in Fig. III.6. There it can shield the telescope from direct light from the star, allowing imaging of the dimmer reflected light from orbiting exoplanets.

The important goal of future direct imaging, with the aid of coronagraphs or starshades, is to allow measurements of the reflected starlight spectra from Earth-like exoplanets. Such studies will enable determination of the chemical composition of the planet’s atmosphere, as well as some information about its surface. Some features one might look for are illustrated in Fig. III.7 by the spectrum of Earthshine. You can see Earthshine by eye because sunlight reflected from Earth faintly illuminates the dark side of a crescent moon, which sunlight doesn’t reach directly. The spectrum in Fig. III.7 has been reconstructed from analyses of that Earthshine on the darker side of the Moon, focusing on the visible and near infrared portions of the spectrum.
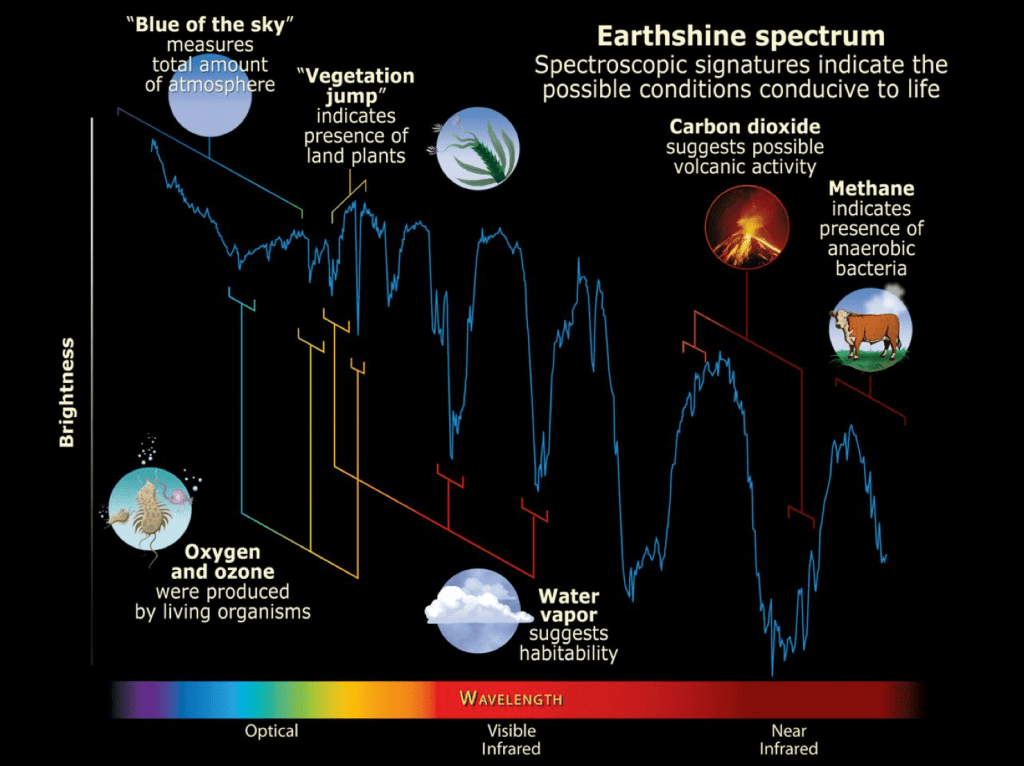
Near the left edge of Fig. III.7, one can detect Earth’s blue sky in the rise in reflectance toward the blue-violet end of the visible light spectrum, which results from sunlight scattering off molecules in the atmosphere. Another rise, labeled in the figure as the “vegetation jump,” results from reflections from plant life on Earth’s surface. This jump is also sometimes referred to as the “red edge,” because the rapid rise is seen at wavelengths somewhat longer than that of green light, since the chlorophyll pigment plants use in photosynthesis absorbs some of the reflected green light. The gradually falling reflectance toward the infrared is interrupted by a series of deep dips, some sharp and some broad, arising from absorption of reflected light by particular molecules in Earth’s atmosphere.
As indicated by labels in Fig. III.7, a set of sharp dips in the visible portion of the spectrum reveal the presence of oxygen and ozone, which, as we noted earlier, are produced by photosynthesizing organisms on the surface. The dips labeled as arising from water vapor indicate the presence of surface oceans. Dips caused by CO2 in the near infrared, as well as ones not shown in the deeper infrared, suggest both volcanic activity and the burning of fossil fuels. A dip at the right edge of the figure and ones deeper in the infrared indicate the presence of methane, some emitted from volcanoes, some from the production of oil and gas, and a lot by anaerobic bacteria and the digestive system of cattle, who emit methane in burps and excretions. The reflected light spectrum thus serves as a “fingerprint” indicating not only conditions favorable to life, but also the existence of life itself, on the planet. As we discuss further in section V, the coexistence of oxygen and methane in a planet’s atmosphere is considered to be a particularly promising signature of life.
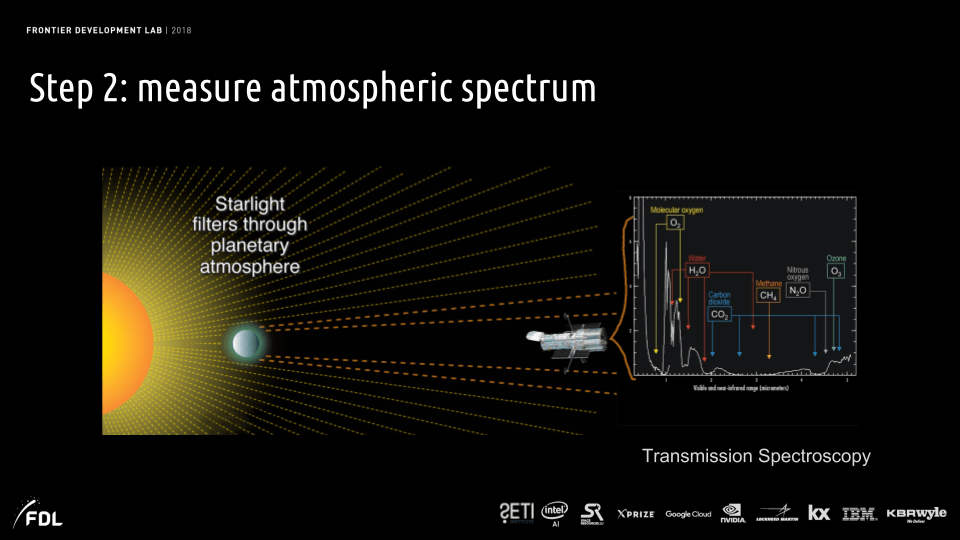
Spectroscopy on the atmospheres of exoplanets can alternatively be studied via light transmission, rather than reflection, when the planet is transiting in front of its star, as indicated in Fig. III.8. Here, one looks for absorption dips in the spectrum of starlight when the line of sight falls in the immediate periphery of a transiting planet. Transmission spectroscopy has been carried out for a number of hot Jupiter-size exoplanets by the Hubble and Spitzer space telescopes. However, dramatic advances in the number and types of exoplanets to be studied with transmission spectroscopy are anticipated with the recently (Christmas Day, 2021) launched James Webb Space Telescope (JWST) pictured in Fig. III.9(a), the largest and most complex space telescope yet developed. An indication of the improvement in resolution with the cryogenically cooled Webb sensors over the earlier Spitzer telescope is shown in Fig. III.9(b). JWST will also be equipped with a coronagraph to perform reflection spectroscopy, as well.

The JWST is an engineering marvel, as can be seen in a video covering some of the details. The engineering challenges are magnified by the fact that JWST can no longer be serviced at its final destination, unlike the Hubble telescope, which orbited Earth. JWST is currently nearly one million miles from Earth, circling a point in space known as Lagrange point 2 (L2). There are five total Lagrange points at which the combined gravitational pulls on an object from the Earth and the Sun are just right to keep the object orbiting the Sun synchronously with the Earth, without the need for additional fuel (beyond the amount it takes to get there). These are shown, along with the JWST trajectory, in Fig. III.10.

A spacecraft like the SOHO observatory positioned at L1, between Sun and Earth, can carry out long-term imaging of the Sun itself. But the JWST at L2 can turn its back to the Sun and the Earth and peer outward into deep space billions of light-years away, shielded simultaneously from infrared background from Sun, Earth and Moon. JWST could not sit for long precisely at L2, because that position is unstable with respect to any kick toward or away from the Earth and the Sun, as might arise from space debris. Instead, it will orbit with a slight wobble around L2 every 6 months (joining the European Space Agency’s Gaia deep space probe already in a different orbit around L2), while the L2 point remains always in Earth’s shadow during its year-long orbit around the Sun. JWST will remain out of Earth’s shadow so that its solar panels can continually power its electronics.
— To be continued in Part II —
