Scientific Tipping Points: The Ozone Layer, Part I
We reviewed the controversy over chlorinated fluorocarbons (CFCs) and the ozone layer in our two-part series on the Ozone Layer Controversy. We urge the reader to read those blog posts; here, we will summarize the major advances in understanding this issue, the negotiations that led to a worldwide ban on CFCs, and we will identify not one but two “tipping points” in the resolution of the ozone layer problem.
At the end of our post we list a number of source documents. A particularly useful document is a summary called Twenty Questions and Answers About the Ozone Layer. It is jointly sponsored by the World Meteorological Organization, the United Nations Environment Programme, the National Oceanic and Atmospheric Administration (NOAA), the National Aeronautics and Space Administration (NASA), and the European Commission. This report is updated regularly and we have used the latest (2014) summary. The book Merchants of Doubt, by N. Oreskes and E. M. Conway, has a comprehensive summary of the ozone controversy in Chapter 4 of that book. Another excellent summary of both the scientific and diplomatic negotiations over the ozone layer is the book Ozone Discourses by Karen T. Litfin (Columbia University Press, 1995). In addition, we thank Prof. Daniel Cole from the Indiana University Maurer School of Law for clarifying some issues regarding negotiations about the ozone layer.
1) The Ozone Layer Hypothesis:
In the early 1970s, atmospheric chemists Sherwood “Sherry” Rowland and Mario Molina became concerned about the behavior of chlorinated fluorocarbons or CFCs in the atmosphere. CFCs are a family of organic compounds that contain at least one chlorine and one fluorine atom, as shown schematically in Fig. 1.1.

CFCs were developed in the 1920s as a result of a search for non-toxic substances that could be used as refrigerants. The boiling points of CFCs made them useful for refrigerants, but many other uses for these chemicals were developed. For example, they were widely used in aerosols; they appeared in foam insulation; they were used as cleansers for electronic components, and products such as polystyrene cups and “packing peanuts” were manufactured in processes that utilized CFCs. The leading manufacturer of refrigerants was the DuPont Company, whose product Freon was widely used as the coolant in air conditioners and refrigerators. By the mid-1970s, around 2 billion pounds of CFCs were being produced each year worldwide.

In 1970, British scientist James Lovelock developed an instrument capable of measuring trace amounts of CFCs in the atmosphere. Lovelock initially measured CFCs in urban areas at levels of parts per trillion. He subsequently measured CFCs over the Atlantic Ocean, suggesting that these compounds were being transported by large-scale air currents.
When Rowland and Molina became aware of Lovelock’s measurements, they began to study CFCs in the atmosphere. They asked the following questions. What happened to CFCs once they entered the atmosphere? Would they degrade and if so through what reactions? Were CFCs accumulating in the atmosphere, and how long would they persist? Would CFCs rise into the stratosphere, and if so what would happen to them there?
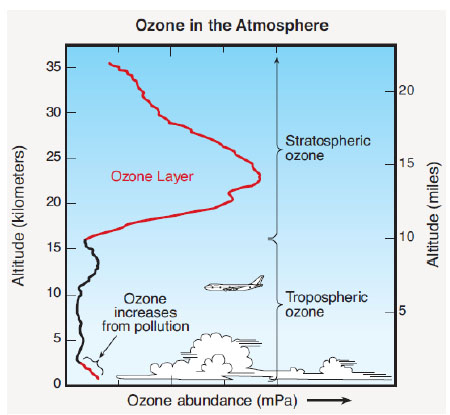
Rowland and Molina discovered that in the lower atmosphere, CFCs could remain for decades, possibly for a century. The chemicals were nearly insoluble in water, resistant to oxidation, and unaffected by sunlight in visible wavelengths. Through mixing with the atmosphere, CFCs would gradually rise into the stratosphere until they reached the level of the ozone layer. Figure 1.3 shows the concentration of ozone vs. altitude. The concentration of ozone in the atmosphere peaks at a height of 20-25 km above the Earth, and this region is called the “ozone layer.”
Ozone is a molecule made up of three atoms of oxygen. Figure 1.4 shows various forms of oxygen: a single oxygen atom; the most common oxygen molecule O2; and ozone O3.

In the ozone layer, ozone molecules absorb deep ultraviolet light from the Sun. This radiation breaks up ozone through the process γ + O3 → O2 + O, where γ represents a UV photon. Ultraviolet light from the Sun is divided into three different categories. UV-C is light with wavelength less than 290 nanometers (nm). UV-C light is lethal to life forms, and 100% of UV-C light is filtered out in the ozone layer. UV-B is light with wavelengths between 290 – 320 nm, and is harmful to most life forms. As a result of the interaction with ozone, most of the UV-B light gets absorbed in the stratosphere and does not reach the Earth, where it would cause skin cancers, cataracts and other environmental damage. UV-A light, with wavelengths between 320-400 nm, is largely unabsorbed in the ozone layer, and much of this light makes it to Earth. At the height of the ozone layer, ozone and O2 are in equilibrium.
Rowland and Molina studied all known reactions of CFCs in the atmosphere. Based on their study they constructed the following hypothesis.
- CFCs would persist in the atmosphere without any reaction breaking them down. They could remain in the atmosphere for up to a century. Because of this long lifetime, CFCs would eventually rise into the stratosphere by mixing with currents in the air.
- Once they reached the upper stratosphere, CFCs would interact with the UV-B radiation present at that altitude. This interaction would release a free chlorine atom.
- The free chlorine atom would break up an ozone molecule, through the reactions (1) Cl + O3 → Cl O + O2; Cl O + O3 → Cl + 2 O2.
These reactions are shown schematically in Fig. 1.5.
- In the reactions of Fig.1. 5, the chlorine atom acts as a catalyst; the chlorine destroys an ozone molecule, but a subsequent reaction between chlorine monoxide (Cl O) and an oxygen atom in another ozone molecule produces a free chlorine. Rowland and Molina calculated that a single chlorine atom freed from a CFC molecule had the potential to destroy up to 100,000 ozone molecules.

In 1974, Molina and Rowland published a paper in Nature [Nature 249, p. 810 (1974)] outlining their hypothesis. The ramifications of their scenario were sufficiently stark that a National Academy of Sciences study was commissioned to examine the potential role of CFCs on stratospheric ozone. The study, completed in Sept. 1976, concluded that the most likely scenario was a depletion of stratospheric ozone levels by 7 — 13% by the year 2050. To put this into perspective, health officials estimated that every 1% reduction in the ozone layer would produce a 3% increase in skin cancers, and it would also produce 100,000 additional cases of blindness caused by cataracts. Rowland and Molina urged the chemical industry and governmental bodies to consider drastic reductions in production of CFCs.
In addition to chlorine, additional catalytic reactions can arise from compounds containing bromine. One common source of bromine compounds is halons, which are widely used in certain types of fire extinguishers. Halons are employed in situations where the use of water might damage sensitive equipment or threaten peoples’ lives. Although halons are produced in much smaller quantities than CFCs, the ozone-depleting potential (ODP) of halons can be up to ten times as large as for CFCs. Although we will mostly confine our discussion to CFCs, the issue of halons and their ODP was discussed in the international negotiations that are described later in this review.
2) Reaction to the Rowland-Molina Hypothesis:
The suggestion by Rowland and Molina was sufficiently striking that the scientific community mounted several parallel efforts to measure CFCs in the atmosphere, and to corroborate (or refute) their hypotheses. First, scientists checked to see if there were reactions in the lower atmosphere that would break down CFCs. At the same time, they mounted efforts to measure CFCs in the stratosphere, and to determine whether chlorine atoms were depleting the ozone layer.
Studies of CFCs and ozone proceeded along three parallel tracks. The first consisted of laboratory measurements of chemical reactions. The second line of investigation involved detailed simulations of the atmosphere. These simulations were designed to predict atmospheric chemical reactions involving CFCs, movements of layers of air, and catalytic reactions involving CFCs and ozone. A third area involved measurements of CFCs and ozone in the atmosphere. Figure 1.6 shows various techniques that are used to measure ozone concentrations in the atmosphere. Some instruments use optical techniques that rely on the Sun or lasers as light sources. A second set of measurements detects thermal emissions from ozone; and a third method relies on chemical reactions that are unique to ozone. One set of instruments measures ozone locally in sampled air, while other techniques measure ozone at remote distances from the instrument.

Not surprisingly, the Rowland-Molina hypotheses were initially opposed by the chemical industry. At that time, as many as 2 billion pounds of CFCs per year were being produced. CFCs generated approximately $8 billion per year in sales, and employed around 200,000 Americans. Particularly for refrigerants, CFCs appeared to be the cheapest and most effective products on the market. So the chemical companies producing CFCs took steps to protect their products. They generated several million dollars in research grants to study the issues raised by Rowland and Molina. In addition, they created lobbying groups that mounted public-relations campaigns on behalf of the industry.
Initially, Rowland and Molina were disappointed at the reaction from their research colleagues. To be sure, their hypothesis was highly speculative and needed to be verified. But some of their fellow researchers had a hard time believing that potentially catastrophic reactions could take place. The atmosphere seemed exceptionally stable, and many scientists assumed that overlooked atmospheric processes would somehow prevent large-scale destruction of ozone.
The skepticism of some chemists was bolstered by the fact that estimates of ozone depletion fluctuated dramatically, as additional chemical reactions were factored into computer simulations of CFCs in the atmosphere. For example, in 1977 the U.S. Congress banned aerosols containing CFCs because of concern over the possible effect on ozone. However, Britain did not support such a ban. British atmospheric chemist John Gribben asserted “On balance, the evidence suggests that nothing mankind is now doing will have a disastrous effect on the ozone layer.” In fact, for a number of years scientists and bureaucrats from the U.S. and the European Community (EC) were at loggerheads over the appropriate response to CFCs and ozone. The Americans argued that the threat from CFCs was substantial and probably required steps to reduce CFC production, while a European faction led by Britain and France minimized any negative effects of CFCs and argued for much less stringent limits on CFCs.
The chemical companies and their consulting scientists mounted a spirited defense of their products. In 1975, DuPont ran full-page ads in newspapers defending CFCs and denying that any hard data connected CFCs with harm to the ozone layer. The Chairman of DuPont stated that Rowland and Molina’s ozone-depletion hypothesis was “a science fiction tale … a load of rubbish … utter nonsense.”
And British Professor Richard Scorer, a chemical industry consultant, asserted that our atmosphere was “the most robust and dynamic element in the environment.” According to Scorer, Rowland and Molina were “doomsayers,” and their entire hypothesis was “pompous claptrap.” The chemical industry argued that CFCs were entering the atmosphere in insignificant amounts, in contrast to volcano eruptions that released vast quantities of chlorine into the atmosphere without causing permanent reductions in ozone.
Another industry tactic was to exaggerate the costs of regulating the chlorofluorocarbon industry. The CEO of CFC manufacturer Pennwalt suggested that phasing out CFCs would cause “economic chaos.” DuPont predicted that economic costs in the U.S. alone could exceed $135 billion and that “entire industries could fold.” The Association of European Chemical Companies claimed that phasing out CFCs could cause “re-design and re-equipping of large sectors of vital industry … smaller firms going out of business … and an effect on inflation and unemployment, nationally and internationally.”
S. Fred Singer, a scientist and senior fellow of the conservative Heartland Institute, claimed that if CFCs were banned, their substitutes “may be toxic, flammable, and corrosive, and they certainly won’t work as well. They’ll reduce the energy efficiency of appliances such as refrigerators, and they’ll deteriorate, requiring frequent replenishment.”
However, experiments soon verified several aspects of Rowland and Molina’s hypothesis. The levels of CFCs in the atmosphere were steadily increasing. And CFCs were incredibly stable – they could persist in the atmosphere for up to a century. Finally, significant indirect evidence suggested that chlorine was destroying ozone; from Eq. (1) and Fig. 1.5 we see that the presence of chlorine monoxide (Cl O) is a “smoking gun” indicating that chlorine is destroying ozone. In 1976, Cl O was discovered in the ozone layer, at levels consistent with the Rowland-Molina hypothesis.
Despite indirect evidence that chlorine from CFCs was destroying ozone in the ozone layer, it proved difficult to obtain direct quantitative proof of ozone depletion. As we have mentioned, CFC concentrations in the atmosphere were extremely small. In addition, there are significant natural fluctuations in ozone concentration from month to month, as well as variations with latitude. The trick was to detect small decreases in ozone levels, and to extract those decreases from a large background of normal seasonal and latitude variations.
By the early 1980s, scientists were attempting to determine precise levels of ozone depletion. And here, two “tipping points” occurred in reasonably rapid succession.
3) Tipping Point #1: The Hole in the Antarctic Ozone Layer
In 1980, the final year of the Carter Administration, the Environmental Protection Agency (EPA) outlined a plan for possible future regulation of CFCs. However, the incoming Reagan Administration was far less welcoming to regulatory action. Reagan’s EPA administrator Anne Gorsuch Burford considered the ozone depletion scenario more of a scare story than reality. And DuPont convened an industry-wide consortium, the Alliance for Responsible CFC Policy, with the stated purpose “to ensure that the government did not regulate based on an unproven and unverified theory.”
The Reagan Administration mandated that any major new regulation be justified on a cost-benefit analysis. Critics of this policy rightly pointed out the difficulty of estimating the costs of actions that had not yet been undertaken. When the Rowland-Molina hypothesis was first published, DuPont was carrying out research on alternatives to CFCs. However, in 1981 two of the largest producers of CFCs, Britain and the U.S., had leaders Thatcher and Reagan who were temperamentally opposed to government regulations on industry. Thus DuPont stopped funding research on CFC alternatives, as it appeared unlikely that restrictions on CFCs were imminent.
In the mid-1980s, the U.S. National Aeronautics and Space Administration (NASA) coordinated a world-wide effort to test the validity of theories that chemicals were depleting atmospheric ozone. A report prepared for the World Meteorological Organization concluded that concentrations of CFCs in the upper atmosphere had doubled in the decade from 1975 to 1985. Furthermore, the report concluded that high concentrations of ozone-depleting substances (ODS) could in principle destroy atmospheric ozone. However, it was difficult to obtain hard quantitative evidence that proved beyond doubt that ODS were destroying ozone. In the early 1980s, there was no “smoking gun” sufficient to justify legislative action on this issue.
Between 1974 and 1987, the Alliance for Responsible CFC Policy requested three more years of research and monitoring before any action was taken on the production of CFCs. During the Reagan years, such requests were routinely approved by EPA officials. Mario Molina referred to this strategy as “the sliding three years.”
It appeared that the net result might be a stalemate, with scientists pressuring for action to limit CFCs while business groups opposed them, while the EPA made it difficult or impossible to impose restrictions on CFCs. However, a new discovery in the mid-1980s would radically change the dynamics of this situation, and tip the scales decisively in favor of action to limit production of CFCs.
Joseph Farman was a British researcher who collected samples from the atmosphere over Antarctica. In 1982, he discovered that ozone abundances had decreased by a shockingly large amount, nearly 40% over a ten-year period. Figure 1.7 shows Farman’s measurements, in units called Dobson Units (‘Dobson Units’ represent a standard measure for atmospheric ozone abundances).

Because of the large seasonal fluctuations in ozone concentration, Farman plotted measurements for one particular month, October. Although the data has considerable scatter (reflecting the significant seasonal and annual fluctuations), there is a noticeable decrease beginning around 1977. After 1977, average October Antarctic ozone levels systematically decrease every year.
Farman’s results from balloon flights over the Antarctic came from a single location, but subsequent measurements from a second site gave the same results. Initially, this was puzzling because NASA satellite data appeared to show no decrease in ozone levels. The disagreement was resolved when it was realized that NASA satellites were programmed to filter out measurements with large deviations from “expected” results. Deviations exceeding 30% were not recorded, because it was assumed they resulted from faulty measurements. When the NASA analysis programs were corrected, their data now showed a massive hole in the ozone layer above the Antarctic, in agreement with Farman’s balloon-based results.
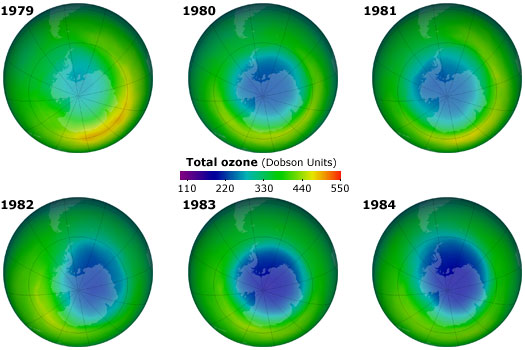
Figure 1.8 shows NASA measurements of October ozone concentrations over the Antarctic as a function of year. Areas shaded in blue represent regions of ozone depletion. The area of the depleted ozone region (roughly the size of the United States) was increasing every year, and the amount of ozone depletion also increased. It was found that the decrease in ozone was largest during the Antarctic spring, with a peak around September or October.
Although scientists were beginning to accept the Rowland-Molina hypothesis of ozone depletion by CFCs, the discovery of the hole in the Antarctic ozone layer dramatically changed the debate. The Rowland-Molina model had predicted ozone depletion at a level of a few percent. In the Antarctic, some hitherto-unknown chemical process was causing ozone depletion at an order of magnitude higher rate than anticipated.
By constructing graphs of the region of the atmosphere above the Antarctic, the “ozone hole” was easily visible (see Fig. 1. 8), and it was straightforward for the general public to comprehend the consequences of ozone depletion. The graphs of the ozone hole over time allowed the public to easily grasp the progressive changes in the ozone layer, in a way that a graph or a table of numbers could not. Since both the area of the depleted region and the extent of ozone depletion were rapidly increasing, this was particularly disturbing to Southern Hemisphere inhabitants in countries such as New Zealand, Australia and Argentina.
The discovery of the ozone hole over the Antarctic, and the plots of ozone depletion shown in Fig. 1.8, marked a “tipping point” in the perception of the ozone issue by both scientists and the public. What had previously been a standoff between scientists and the chemical industry rapidly shifted to a situation where the public demanded action to preserve the ozone layer. These demands were supported by a significant majority of the scientific community.
But before a plan to save the ozone layer could be formulated, it was necessary to understand why Antarctic ozone levels were decreasing so rapidly. Susan Solomon from the National Oceanographic and Atmospheric Administration (NOAA) speculated that the decrease in ozone could be related to the presence of high-altitude clouds of ice particles that formed over Antarctica during its winter season. She and colleague Rolando Garcia constructed an atmospheric model that included ice particles. Their model predicted that the solid surfaces of ice crystals would greatly increase the rate of destruction of ozone by CFCs. Collaborating with Rowland and Molina, Solomon showed that chlorine would destroy ozone at a much faster rate in the presence of ice surfaces.
On ice surfaces, the mechanism for ozone destruction was different from that shown in Fig. 1.5. However, the net effect was the same: ozone was converted to oxygen in the presence of molecules containing chlorine, and the process was catalytic. In the Antarctic, stratospheric clouds containing ice crystals only form during a six-month period from May to October, when the temperature is sufficiently low. Furthermore, the catalytic process that destroys ozone requires visible sunlight. September and October are the only months with both low Antarctic temperatures and sufficient sunlight to enable the catalytic process.
A second factor in the depletion of Antarctic ozone was the role played by the polar vortex. The polar vortex is caused by powerful winds that circulate around each pole, and that are strongest during the winter season. The polar vortex confined the ozone-depleted region so that it remained over the Antarctic. In the absence of the polar vortex, air from mid-latitudes containing undepleted ozone would mix with Antarctic air, diminishing the magnitude of the Antarctic ozone hole.
Once the magnitude of the Antarctic ozone hole was precisely measured, and the mechanism that caused the ozone decrease was understood, a strong consensus developed that the decrease in ozone levels represented a crisis that demanded concerted action.
4) Tipping Point #2: DuPont ‘Flips’ Its Position On CFCs
In Section 2 we noted that the DuPont company, holder of the patent for Freon and the world’s largest manufacturer of CFCs, was initially dismissive of suggestions that CFCs might be responsible for depleting the ozone layer. For example, following the hypothesis by Rowland and Molina that chlorine from CFCs might migrate to the stratosphere and destroy ozone, DuPont ran ads emphasizing that no hard data demonstrated any harm to the ozone layer from CFCs. DuPont spokespersons predicted that a ban on CFC production would entail enormous economic costs and that “entire industries could fold.”
However, in 1974 the Chairman of DuPont, Irwin Shapiro, pledged that his company would cease production of CFCs if it was shown that they endangered human health. On the other hand, The Alliance for Responsible CFC Policy, an industry-wide consortium led by DuPont, had been convened as a means of preventing government regulation of CFCs. So between 1974 and 1986, DuPont was simultaneously cultivating an image as a responsible corporation that took action to minimize health issues resulting from their products, and bankrolling a consortium whose goal was to prevent government regulation of their products.
In 1985, the United Nations Environmental Programme established the Vienna Convention for the Protection of the Ozone Layer. When the Vienna Convention was first established, the severity of ozone depletion remained in dispute. In particular, there was a disagreement between the U.S., which was pushing for rapid reductions in CFCs and other ozone-depleting substances (ODS), and the European Union, which maintained there was insufficient evidence of ozone depletion to require any immediate restrictions on CFCs and other ODS.
However, the Vienna Convention did provide a framework for further negotiations. As discussed by Daniel H. Cole in Climate Change and Collective Action, the parties “agreed to support further scientific research, cooperate on efforts to resolve scientific, technical and legal issues, and participate in a ‘conference of the parties,’ which included regularly scheduled meetings. The Convention created a ‘secretariat’ to facilitate negotiations and oversee cooperative efforts. Most importantly, the framework convention established procedures for adopting binding protocols and amendments. Finally, the framework convention provided procedures by which the Convention itself and future protocols would be voted upon, ratified and implemented.”
The initial 1985 Vienna Framework Convention did not impose restrictions on CFCs, but it established a governance structure that allowed it to monitor and react to future information regarding ozone depletion. Therefore, a Vienna Convention working group met to discuss the recent report from the British Antarctic Survey announcing a hole in the Antarctic ozone layer.
In 1986, following the discovery of the Antarctic ozone hole, DuPont announced that they could support restrictions on CFCs; however, they insisted such restrictions would have to be international and not a unilateral U.S. decision. American chemical manufacturers felt that the 1979 U.S. ban on aerosols put them at a disadvantage relative to international firms that continued producing aerosols.
DuPont and other chemical manufacturers were aware that discovery of the Antarctic ozone hole had completely changed the public perception of CFCs. If CFCs were destroying the ozone layer, the number of skin cancers would rise dramatically. This would leave companies producing CFCs at risk of lawsuits for harming human health. Furthermore, DuPont had underwritten research that produced state-of-the-art simulations of CFCs in the atmosphere, and those simulations strongly suggested that CFCs were responsible for the observed ozone depletion.
Following the discovery of the Antarctic ozone hole, scientists focused on ozone levels at mid-latitudes. No reduction in ozone levels had been detected there, despite the fact that ground-based measurement stations had been tracking ozone levels around the globe since the 1950s. Solomon’s ice-cloud model predicted that there would also be seasonal reductions in Arctic ozone levels, although Arctic ozone depletion was predicted to be much smaller than the Antarctic. To understand these apparent discrepancies, in 1987 NASA organized the Ozone Trends Panel, consisting of 150 scientists from around the world.
The Ozone Trends Panel re-evaluated the ground-based data and concluded that there was an average ozone depletion of 1.7 – 3% in the Northern Hemisphere, and up to 6% in winter. This had not been observed earlier, because previous studies assumed that ozone levels would be the same throughout the year and at all latitudes. Thus, measurements taken at different times of year and different latitudes were lumped together and averaged. Now it appeared that ozone was being depleted at all latitudes, in agreement with simulations that combined the Rowland-Molina mechanism with the model of Solomon.
A DuPont scientist, Mack Macfarland, was a member of the Ozone Trends Panel. He had read a draft of the Panel’s report before it was issued on March 21, 1988. Macfarland contacted DuPont’s head of government relations and informed him that the connection between CFCs and ozone depletion had been verified. As a result, in 1988 DuPont announced that they would voluntarily cease production of CFCs within approximately ten years. DuPont also persuaded other American CFC manufacturers to support international regulations on CFC production.
The about-face by DuPont on CFCs was a major step towards an eventual world-wide ban on CFC production. DuPont used its position as the world’s largest producer of refrigerants to convince other companies to join them. Initially, there was considerable friction with other chemical companies; some accused DuPont of pulling the rug out from under the industry Alliance. Others were convinced that DuPont had secretly developed replacements for CFCs, and that their announcement was a sign that DuPont intended to dominate licensing and production of CFC alternatives.
At the end of this post, we will review questions regarding DuPont’s actions during this period. However, regardless of their motives, there seems little doubt that DuPont’s public announcements (in 1986, that they were willing to consider restrictions on production of CFCs; and in 1988 that they would phase out production of CFCs) marked a second “tipping point” leading to the regulation of CFCs and ODS
5) A Ban on CFC Production.
The 1985 discovery of the hole in the Antarctic ozone layer sparked a call for action on CFCs. After two years of negotiations, the Montreal Protocol on Substances that Deplete the Ozone Layer was agreed to in 1987. In the Montreal Protocol, the original signatory countries agreed to consider reductions in the production of CFCs, and they agreed to meet at regular intervals to review and update the status of ozone in the stratosphere.
In 1988, the Ozone Trends Panel reported their results. They found ozone depletion in the mid-latitudes in winter. In May 1988, at the time when the Ozone Trends Panel released its findings, the U.S. Senate voted to ratify the Montreal Protocol. That Protocol went into effect in Jan. 1989.
When DuPont announced in 1986 that it was willing to consider restrictions on CFC production under an international agreement, it initially envisioned either a “freeze” on CFC production at the current level, or a phase-out of CFC production over a relatively long time scale. This would allow time to search for CFC alternatives, and would also provide manufacturers with a significant window to phase out CFCs and replace them with alternatives. However, studies revealed that ozone was being depleted at a significantly higher rate than anticipated, so the timetable for phase-out of CFCs was shortened. Prodded by DuPont, the Alliance For Responsible CFC Policy eventually supported restrictions on CFC production.
In 1990, the Montreal Protocol was amended to call for complete elimination of CFC production by the developed countries by the year 2000, with elimination from developing countries by 2010. An important aspect of the Montreal Protocol was that it included a $400 million fund to assist developing countries cover their costs of compliance. Over the years, more and more countries ratified the Montreal Protocol. At present, every nation in the world (196 of them) has signed the Montreal Protocol; it is the only environmental treaty ratified by every nation.
The Montreal Protocol was an historic document: it “was the first international treaty to address a global environmental regulatory challenge; the first to embrace the ’precautionary principle’ in its design for science-based policymaking; the first treaty where independent experts on atmospheric science, environmental impacts, chemical technology, and economics, reported directly to Parties, without edit or censorship, functioning under norms of professionalism, peer review, and respect; the first to provide for national differences in responsibility and financial capacity to respond by establishing a multilateral fund for technology transfer.”
The chemicals currently used as CFC replacements for refrigeration are hydrochlorofluorocarbons (HCFCs), compounds that are more reactive than CFCs. HCFCs break down in the atmosphere much faster than CFCs, and thus are significantly less likely to reach the stratosphere. As a result, although HCFCs still have the potential to deplete the ozone layer, ozone depletion from HCFCs is significantly smaller than the hazard from CFCs.
Fig. 1.9 lists the ozone-depletion potential (ODP) for a number of CFCs, HCFCs and HFCs (chlorine-free hydrofluorocarbons). The ODP is calculated relative to the compound CFC-11 which is assigned a value 1. Note that the HCFCs generally have ODP substantially smaller than CFC-11, and that the HFCs have zero ozone-depleting potential. Note also that bromine compounds, which are prevalent in gases called halons, have very high ODPs. Halons are widely used in commercial and military fire-extinguishing systems.

However, because HCFCs still degrade the ozone layer to some degree, they are considered only a temporary replacement for CFCs. The next substitute for CFCs will be the HFCs, which do not destroy stratospheric ozone at all. In 2007, representatives from 196 countries met in Montreal in a summit sponsored by the United Nations. They agreed to a timetable that would eliminate hydrochlorofluorocarbons entirely by 2020, with developing nations allowed an extension until 2030.
However, an additional consideration for both HCFCs and, especially, HFCs is that they are rather potent greenhouse gases. The relative global warming potential (GWP) of each compound is shown in the right-hand column of Fig. 1.9. Thus, in addition to concerns for the ozone layer, one has to consider the contributions of replacement gases to global climate change. From Fig. 1.9, note that some HFC compounds have quite low GWPs. At present, countries are beginning to use hydrocarbons (particularly a blend of propane and isobutane) as replacements for HFCs, as they have negligible environmental impact. Hydrocarbons are being increasingly used in countries such as Australia and the U.S., particularly for mobile air conditioning units.
— To Be Continued in Part II —
