Note added June 24, 2023: The U.S. is adding two new pressurized water nuclear reactors, Vogtle-3 and Vogtle-4, to an installation near Waynesboro, Georgia. Each of these plants will provide 1,100 Megawatts of power. The two plants are the first Generation III nuclear reactors in the U.S., and the first new nuclear power plants constructed in the U.S. since the 1979 level 5 incident at Three Mile Island. The reactor design is called AP1000 (for “advanced passive”), and Generation III plants have a number of features that are touted to increase the safety of the reactors. First, AP1000 reactors have 50% fewer safety-related valves, 35% fewer pumps, 80% less safety-related piping, 85% less control cable, and 45% less seismic building volume than earlier U.S. reactors. The design allows water passing through the core to rise as it heats; and if the reactor leaks or loses power, then emergency cooling water is designed to flow from a tank located above the reactor core. The water flow begins by using explosively operated and DC operated valves, which operate independently of operator control; the water then falls by gravity. These passive features a claimed to be significantly safer than pumping systems used in earlier Generation II reactors. Another safety feature is that these reactors include a “core catcher.” In the event of a serious accident where the fuel rods melt, the molten material will fall into the core catcher at the bottom of the reactor vessel. It is claimed that this will prevent the possibility of molten radioactive material (called “corium”) from melting through the bottom of the reactor vessel and being released into the environment. The Vogtle-3 reactor reached full power on May 29, 2023, and the identical Vogtle-4 reactor is expected to attain full power later in 2023.
Note added August 13, 2022: We have updated the parts of this post with a number of minor edits to incorporate some suggestions made to us by Tom Cotton (not the Senator), formerly of the U.S. Office of Technology Assessment. We are grateful to Cotton for his careful reading of our posts and for his constructive comments.
July 26, 2022
I. introduction
Burning fossil fuels has provided most of the energy needed to power rapid expansions in global population and global quality of life over the past century and a half. But their contributions to the rapid increase in the concentration of greenhouse gases in Earth’s atmosphere, to global warming, and to consequent climate change, are by now very clearly established, even if still subject to some quantitative uncertainty. A rapid transition away from carbon-based energy production must occur over the coming decades if the world’s population is to avoid the upheavals accompanying very harsh and painful adaptations to a warmer climate.
Renewable sources of power, especially wind and solar power, have made rapid progress over the past decade and will certainly comprise significant parts of that transition. But there are legitimate questions about whether wind, solar, hydroelectric and biomass power will provide sufficient, and sufficiently steady, output to fuel all the energy needs of a still expanding global population and anticipated rapid economic growth in many developing countries. It is thus essential to consider what the role should be over the remainder of this century for nuclear power, which is certainly carbon-free but hardly free of challenges on technical, health, management, oversight, and public perception grounds.
Nuclear power comes in two different forms, based on the fission of heavy nuclei and the fusion of very light nuclei. The challenges facing these two forms are quite different. In the case of fission power, past reactor accidents at Three Mile Island, Chernobyl and Fukushima have eroded public support, and repositories for long-term radioactive waste disposal have still not been identified in the U.S. and most other countries. Public fear of long-term health effects of radiation remains strong and is very understandable. The focus in the nuclear energy sector recently has been on designing more, smaller power plants – so-called modular nuclear reactors – for the future.
Fusion power – trying to produce energy on Earth with the same basic approach that the Sun and other stars use internally – is carbon-free, relies for the most part on abundant fuel (hydrogen isotopes), produces little radioactive waste, and will not lead to meltdown. But it is technically extremely challenging to generate more energy than one uses in generating it. Throughout our professional careers, fusion power has been projected to become economically viable in about 50 years, but that 50-year projection has remained essentially constant despite 50 years passing. However, there is now a renewed sense of enthusiasm for fusion resulting from three recent developments: technical breakthroughs achieved in two distinct approaches to harnessing fusion; the progress on an enormous fusion research reactor funded jointly by 35 countries; and an influx of substantial private investment in a significant number of commercial companies exploring diverse methods for generating fusion power.
This post has four parts. In Part I, we describe some of the basic physics behind fission and fusion energy production and review basic elements of fission reactor design. In Part II, we review the causes and aftermaths of the three most serious past fission reactor accidents. In Part III, we describe basic elements of a few different types of small modular fission reactor design being evaluated for future nuclear power installations. In Part IV, we review recent progress on attempts to harness fusion power and we assess the possible roles of both fission and fusion technologies in the needed 21st-century transition in worldwide power production.
II. the basic science of nuclear fission and nuclear fusion
The microscopic basis for nuclear power lies in nuclear reactions that release significant amounts of energy. We can understand this energy release by utilizing Albert Einstein’s mass-energy equation E = mc2. In this equation, “c” represents the speed of light, 300 million meters per second. In the present context, “m” is the change in mass in a reaction (the initial mass minus the final mass). When m is positive, E is the energy that is released. If m is negative, E is the amount of energy we must provide to make the reaction proceed.
For example, a proton and an electron form a bound state, the hydrogen atom. When the electron is in its lowest-energy state in hydrogen, the binding energy is 13.6 electron-Volts (eV, a convenient unit of energy for atomic systems), equivalent to the motional (kinetic) energy an electron would gain if it were accelerated across a voltage of 13.6 volts. The mass of the hydrogen atom (mH) is less than the sum of the masses of a proton (mp) plus an electron (me): Δm = mp + me – mH = 13.6 eV/c2. To put this another way, if you take a hydrogen atom in its lowest-energy state, and you pull apart the proton and electron so that they are essentially an infinite distance from one another and at rest, you will have to expend 13.6 eV of energy to accomplish that task.
Except for hydrogen, whose nucleus consists of a single proton, all nuclei are composed of neutrons and protons (collectively called “nucleons”). But, in analogy to the hydrogen atom example, the mass of stable nuclei is always slightly less than the sum of the masses of all the neutrons and protons inside it, because all those nucleons are bound together by the attractive nuclear force. The energy associated with that binding, divided by c2, subtracts from the total mass of the nucleus. That binding energy varies as a function of the number of nucleons A in a well-understood and very well-measured way, as reflected in Fig. II.1. Note that the binding energies in Fig. II.1 are specified in millions of electron volts, as opposed to the tens of eV typically involved in binding electrons to nuclei in atoms.
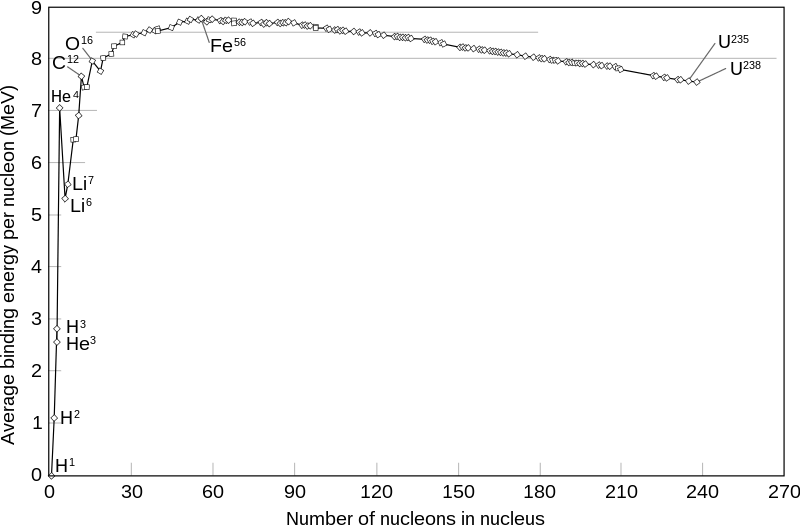
As one moves from hydrogen at the left end of Fig. II.1 up toward oxygen-16, the average binding energy per nucleon increases rapidly because each nucleon is gaining nearest neighbors to interact attractively with. The binding energy per nucleon continues to increase, but more slowly, up to a maximum at iron-56. Beyond iron, the increasing electric repulsion of all the positively charged protons stuffed inside the small nuclear volume counteracts the attractive nuclear force to an increasing extent, so that the net binding energy per nucleon decreases gradually toward uranium. The overall trend is mostly smooth, but is interrupted by occasional minor or major spikes. The most notable spike in Fig. II.1 occurs for helium-4, which is anomalously tightly bound because its configuration of two protons and two neutrons completes the first nuclear “shell,” below an energy gap, for both protons and neutrons. Nuclei with closed shells for both protons and neutrons are dubbed “doubly magic.” The next doubly magic nucleus is oxygen-16, with eight protons and eight neutrons, and a small spike in the figure.
Nuclear reactions conserve the total number of nucleons, as long as anti-protons and anti-neutrons are not involved, although neutrons may change into protons, or vice-versa, in some reactions and radioactive decay processes. Thus, except for the small mass difference between protons and neutrons, the energy release in nuclear reactions is mostly determined by the change in the average binding energy of the nucleons between the interacting nuclei and their reaction products. Reactions that lead toward the maximum at iron-56 in Fig. II.1 gain nuclear binding energy, hence, they lose mass and (again, by Einstein’s equation) convert that lost mass to energy of motion (kinetic energy) of the outgoing product nuclei. Fission reactions release energy by splitting heavy nuclei, like uranium, into lighter nuclei. Fusion reactions release energy when two of the lightest nuclei combine together to produce a nucleus with more nucleons and more binding energy per nucleon. And the fusion energy gain is especially large when the final product is helium-4, since that nucleus is anomalously tightly bound.
Uranium-235 (U235), despite the fact that it would have a great deal of energy to gain, seldom fissions spontaneously. This is true because as a uranium nucleus elongates in shape along a path toward fragmentation into smaller parts, its surface area increases, more nucleons find themselves with fewer nearest neighbors, and the average binding energy decreases rather than increases. The net result is that the uranium nucleus has to pass through a configuration of higher mass than its initial value, creating a small energy barrier it must get over to realize the much larger mass reduction when it splits completely (see Fig. II.2). (For some heavy nuclei there may even be a multi-humped barrier.)
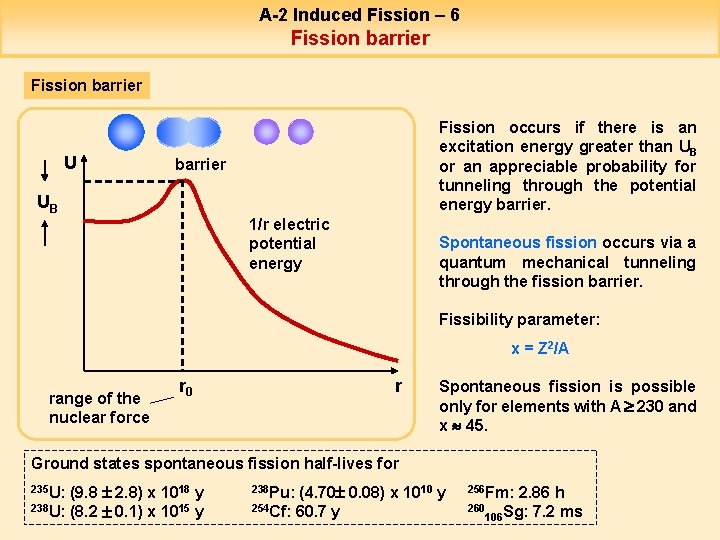
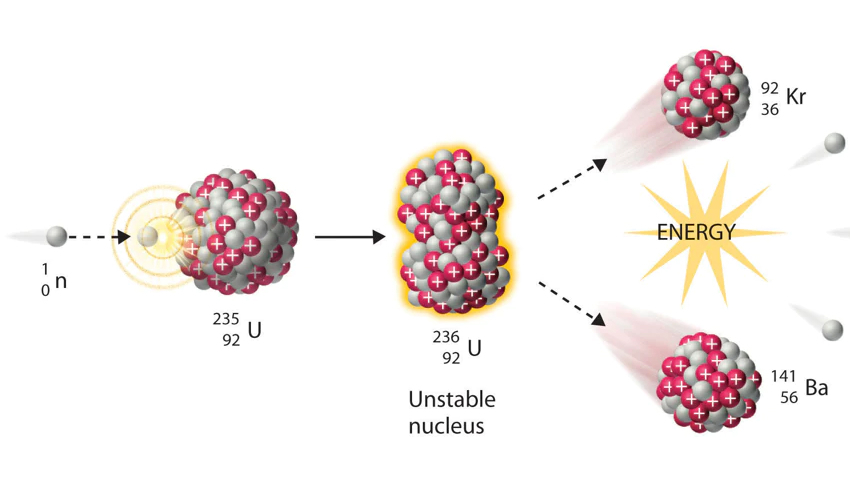
Bombardment of the uranium nucleus with a neutron will provide the boost to get the nucleus over that fission barrier. And when the uranium nucleus splits, it emits typically two or three more neutrons in addition to its larger fragments and nearly 200 MeV of kinetic energy shared by the products. For example, a typical fission reaction resulting from neutron bombardment of U235 is shown in Fig. II.3, resulting in the two lighter nuclei Kr92 and Ba141 plus three neutrons. The basic reason that more neutrons are emitted in fission reactions is that lighter nuclei have a smaller ratio of neutrons to protons than the heavy fissioning nucleus itself.
If you collect together a critical mass of uranium (52 kilograms for a sphere of uranium-235) and initiate the first few fission reactions, the emitted neutrons will cause further nuclei to fission, emitting still more neutrons and causing still more fission, and so on. One thus gets a self-sustaining chain reaction, which is at the heart of fission reactors, as well as of atomic bombs. The major challenges for a fission reactor are to maintain control of the chain reaction, to transport the enormous heat generated away from the fuel and convert it efficiently to electricity, and to deal with ultimate disposal or storage of radioactive product nuclei, some of which are quite long-lived. Control is aided by the fact that not all of the neutrons from each fission reaction are emitted promptly; some result after some delay from the radioactive decay of product nuclei. In an atomic bomb, the prompt neutrons alone are sufficient to fuel the chain reaction and a rapid, powerful explosion results. In a reactor, both the prompt and delayed neutrons are needed to keep the chain going, so that operators and safety systems have a little time to react to threatening conditions.
The Sun and other stars produce copious energy by fusing hydrogen nuclei in a chain of fusion reactions illustrated in Fig. II.4. The net result of that sequence of reactions is that four protons combine to form a single helium-4 nucleus plus two positrons, two neutrinos, and two gamma rays, releasing a total amount of energy equal to 26.7 MeV. The energy release per nucleon involved is much greater than in fission, because the slope at the left edge of Fig. II.1 is much greater than on the right side of the plot. But the cumulative energy release is smaller than in a fission reaction because there are many fewer nucleons involved. On the other hand, unlike uranium and other very heavy elements, hydrogen is the most abundant element in the universe, produced in the early aftermath of the Big Bang.
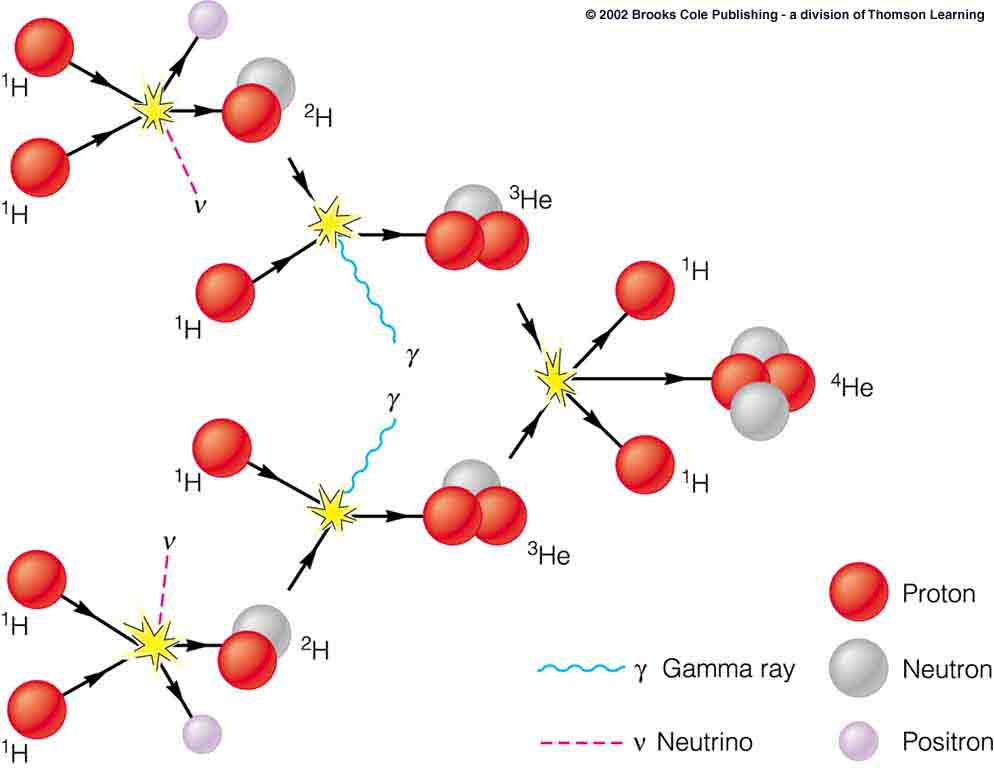
However, it requires extreme density and temperature to make the fusion reactions of Fig. II.4 proceed at significant rates. Protons and light nuclei need to come into very close contact in order to fuse. But they are all positively charged and repel each other electrically. That electric repulsion can be overcome if the nuclei move around with sufficient velocity, and the kinetic energy that accompanies that velocity is available at very high temperatures. That is why the energy production process in stars is referred to as thermonuclear fusion. It requires a very substantial initial input of energy from a source other than fusion to heat a significant mass of hydrogen to the temperatures necessary for fusion to begin. In stars that initial energy input comes from the gravitational attraction that brings all that hydrogen together. In the case of the Sun, that gravitational attraction raises the core to a temperature of about 15 million °C, a temperature that is then maintained by a fraction of the energy produced in the fusion reaction chain.
The inward pressure from all the mass accumulated in the Sun leads to a central pressure of 3.84 trillion pounds per square inch, or some 260 billion times greater than atmospheric pressure at Earth’s surface. Although the Sun is gaseous, and we tend to think of gases as having low density, this enormous pressure leads to a core density of the Sun of about 150 grams per cubic centimeter, or 150 times the density of water, 13.3 times the density of lead. At that central density and temperature, hydrogen atoms are ionized and the matter at the core is a hot plasma comprising mostly free protons and free electrons.
The protons are packed together so tightly in the Sun’s core that there is only about 0.03 nanometers separation, on average, between nearest proton neighbors. At the Sun’s central temperature, the mean thermal kinetic energy of each proton is roughly 2 keV, or equivalent to the energy a proton would gain by being accelerated through a potential difference of 2000 volts. Under those conditions, the closely packed, rapidly moving protons can fuse efficiently. If you burn all the hydrogen contained within a cube with a side length of 1 centimeter, you then generate 9.6 x 1013 Joules, or enough to power 30,000 100-watt light bulbs for a year, if you could capture it all. In the Sun that released energy maintains the ultra-high internal temperature and also generates the enormous energy the Sun emits outward to the solar system.
The challenge, then, for producing thermonuclear fusion energy in power plants on Earth is finding ways to raise light-nucleus fuel to such enormous temperatures and/or densities that fusion can proceed at sufficiently high rates to deliver back more energy than one puts in to create the necessary conditions. In addition, one must come up with ways to confine a plasma containing the fusing nuclei so that it does not remain in contact with other materials, which would melt at the required temperatures. In a hydrogen bomb the initial energy input is provided by first setting off a fission bomb, but that approach is clearly not adaptable to a power plant setting where one wants controlled thermonuclear fusion.
Having explained the basic challenges, we now proceed to review the history of attempts (and failures) to generate energy reliably and cost-effectively in fission and fusion reactors, with greatest emphasis on current research and the outlook for each type of nuclear energy.
III. Fission Reactor design and challenges
Nuclear power plants to date have all been based on fission of heavy nuclei, mostly of the uranium isotope U-235. For most reactor designs, one first has to enrich naturally occurring uranium, in which U-235 accounts for only 0.7% of the uranium. The heavier isotope U-238 accounts for 99.3%, but that isotope does not undergo fission. The enrichment of uranium typically is accomplished using centrifuges. A gas centrifuge used to separate the different isotopes of uranium is shown in Fig. III.1. A gas (generally uranium hexafluoride) is pumped into the centrifuge. When the centrifuge spins, the force on the gas molecules is proportional to the mass of the molecule. Thus, after spinning the centrifuge, the heavier molecules of U-238 will be slightly more prevalent on the outer edge of the centrifuge, while the lighter U-235 will tend to be in the center.
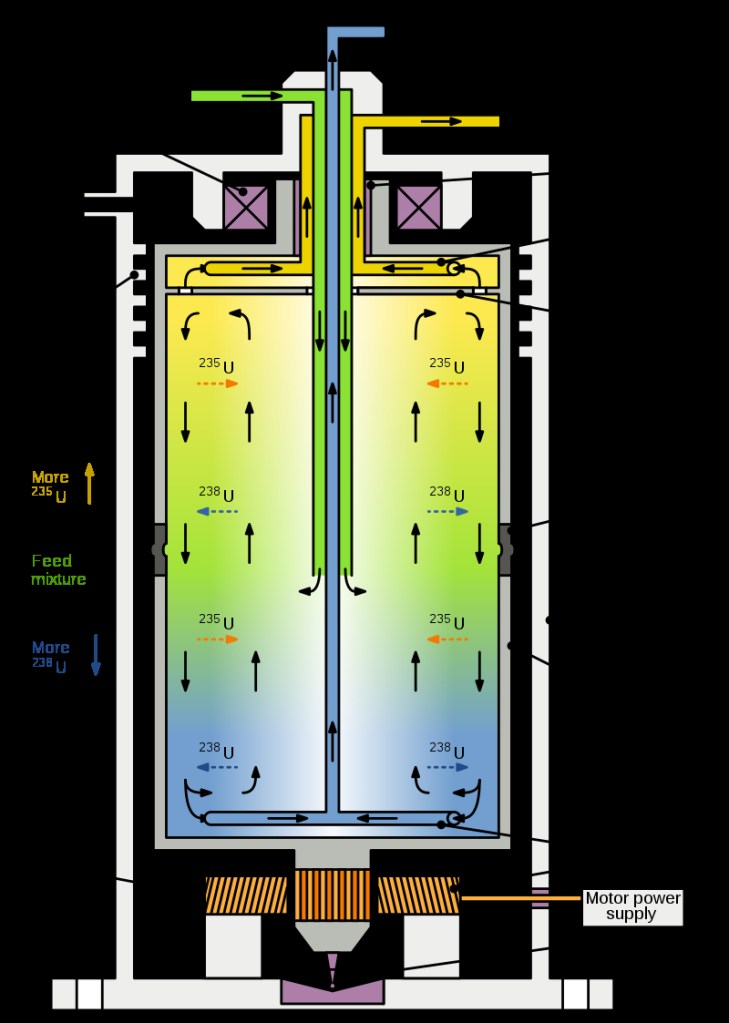
The centrifuges used in separation of uranium isotopes are very large (about 12 meters high), and spin at extremely high rates. After spinning in the centrifuge, the gas is then separated into two parts, where the part closer to the center of the centrifuge will have slightly larger abundance of U-235 than the original gas. The centrifuge process is repeated many times; eventually, one ends up with a substance that has a high enough fraction of U-235 to be used in fission reactors.
To release as much energy as one gets from the fission of one kilogram of U-235, one would have to burn 2.7 million kilograms of coal! The International Atomic Energy Agency currently reports a total of 441 fission power reactors in operation around the world, cumulatively producing nearly 400 billion watts (or 400 GW) of power, with another 53 power reactors currently under construction. Nuclear power plants produce roughly 10% of the world’s electricity. In addition, there are more than 800 fission reactors worldwide that are used for scientific research rather than power generation. The power reactors come in several different versions, but we can illustrate the basic principles of operation by describing the most common type, the pressurized water reactor (PWR).
Figure III.2 illustrates the basic elements in the core of a PWR. The fission fuel is contained in a set of typically vertical fuel rods. The fuel itself generally comprises ceramic pellets of uranium oxide, enriched to increase the abundance of the isotope U-235 from its natural value of 0.7% in mined uranium up to as much as 5% (so called low-enriched uranium, whose enrichment is too low to produce a nuclear explosion). The pellets, which typically have sizes comparable to a sugar cube, are packed in long tubes constructed from a zirconium alloy that is corrosion-resistant and absorbs very few of the neutrons produced in the fission reactions. A PWR producing output electrical power of 1 gigawatt might contain 51,000 fuel rods with over 18 million uranium oxide pellets inside them, providing nearly 500 kilograms of uranium.
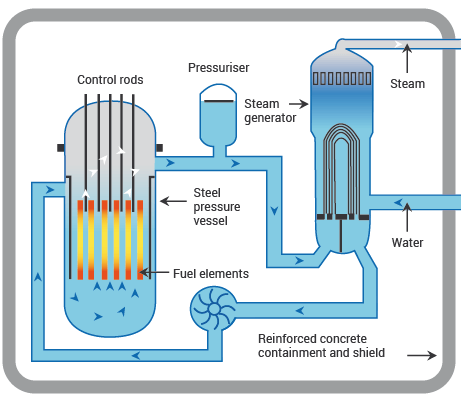
The fission chain reaction is typically launched by neutrons emitted from a radioactive source that contains beryllium in combination with a heavy radioactive nucleus, such as radium or polonium. When the heavy nucleus decays, it emits a helium-4 nucleus (or alpha particle), which combines with a beryllium nucleus to produce carbon-12 plus a neutron. But once the chain reaction has been initiated, the important tasks are controlling it, removing the enormous heat generated in the vicinity of the fuel rods, and converting that heat efficiently to electric power. The water in a PWR plays an essential role in all of these tasks.
Water acts as a moderator, reducing the average energy of the neutrons emitted in uranium fission by having them collide with multiple protons in the water molecules. The moderation yields neutrons that are more efficient in inducing U-235 fission, and less efficient in producing the more fissile nucleus plutonium-239 by absorption on the more abundant uranium isotope U-238. (Alternative designs known as fast neutron reactors or fast breeder reactors use a liquid other than water in the core, in order to reduce moderation and focus on plutonium production and fission.)
Control of the rate at which the fission chain reaction proceeds is governed by the remote insertion or withdrawal of the control rods pictured in Fig. III.2. These are constructed from materials – for example, cadmium, hafnium or boron – that are especially efficient at absorbing neutrons before they can induce further fission reactions. When a reactor starts up, the control rods are initially inserted all the way and no fission occurs. The control rods are then slowly pulled outward from the reactor core. As the control rods are withdrawn, the rate of the fission process increases, as more neutrons survive to initiate fission reactions. The position of the control rods can be changed to insure that on average, one neutron survives to produce a subsequent fission reaction. If the fission process proceeds too rapidly, the control rods are inserted further into the reactor core to allow fewer neutrons to create fission. The goal in a power reactor is to maintain conditions such that the delayed neutrons resulting from radioactive decay of fission products are needed, in addition to the prompt neutrons, to keep the chain reaction going. Those conditions offer operators and safety systems more time to regain control or halt the chain reaction when something goes wrong.
Water circulates throughout the PWR also to act as a coolant, maintaining a steady temperature in the reactor core despite the enormous heat generated by fission products. In a PWR that temperature may reach 325°C, but the water in the core is kept from boiling by maintaining it under very high pressure, about 150 times atmospheric pressure, in a robust steel vessel containing the fuel and control rods. Water exiting the steel vessel at very high pressure and temperature then enters a separate vessel, where it transfers its heat to a cooler water reservoir, in order to generate steam. The fuel vessel and the steam generation vessel are enclosed in meter-thick containment walls typically constructed from concrete and steel. The containment walls are intended, among other purposes, to keep radioactive fuel and fission products from exiting the power plant.
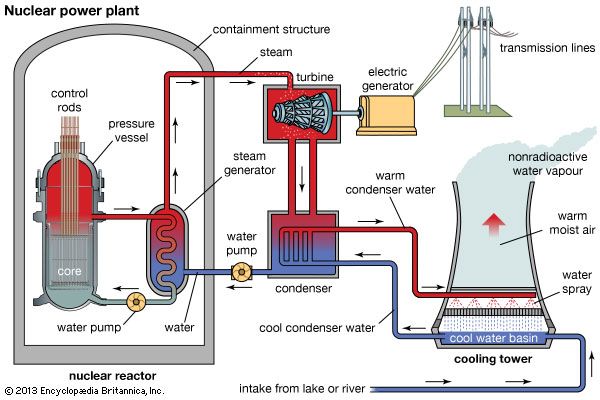
The steam generated inside the containment walls exits through a pipe to the exterior to rotate turbines that run electric generators. A more complete picture of a PWR plant, including the elements outside the containment structure, is shown in Fig. III.3. Note in this figure that the hot pressurized water circulating from the reactor core runs through pipes in the steam-generating vessel, so that if any radioactivity is contained within that hot, high-pressure water, it should not contaminate the cooler, lower-pressure water that generates steam exiting the containment structure. In addition to the electricity generation, this figure also illustrates the cooling tower, with intake from a nearby body of water, and the condenser that reliquefies entering steam to return warm water to the cooling tower and complete the water circuit. Note in Fig. III.3 that there are three distinct water circuits: one involving the water that directly cools the fuel rods; a second processing the water used to generate steam; and a third that supplies water through the heat exchanger in the condenser.
A number of pressurized water reactors first designed in Canada (and known as CANDU reactors) use heavy water, in which hydrogen is replaced by its heavier isotope deuterium in the water molecules, as the moderator and coolant. Heavy water is a more efficient moderator than normal water, and it allows the use of less expensive naturally occurring uranium, rather than enriched uranium, in the fuel rods. A side benefit of the CANDU reactors is that fission neutrons reacting with the deuterium produce tritium as a “waste” product. Although it is itself radioactive, tritium is not ordinary radioactive waste; it is highly valued as an element of the fuel for fusion reactors, as we discuss later in Part IV of this post. At the moment, the CANDU reactors are the most important sources of tritium worldwide.
Under normal operations, fission power plants efficiently produce large amounts of power without burning fossil fuels or increasing the amount of carbon dioxide in the atmosphere. In addition, reactors contain multiple “fail-safe” layers of protection, designed to prevent the reactor from ever cycling out of control. However, there are two major sources of problems associated with fission power production. The first problem is the issue of radioactive nuclear waste. And the second issue is potential problems if the reactors fail to operate normally, and the fail-safe procedures somehow fail to work.
Radioactive waste:
Most power reactors are shut down temporarily for maintenance every year or two, to replace a fraction of the fuel rods with fresh ones. In the rods that have been “used up” or “spent,” much of the U-235 has already undergone fission, and a number of the lighter fission product nuclei are neutron absorbers that reduce the fuel’s effectiveness in sustaining a controlled chain reaction. 96% of the remaining (non-oxygen) mass in a spent fuel rod typically consists still of uranium, but mostly U-238. Roughly 1% comprise plutonium isotopes formed from neutron absorption on U-238, and these products are carefully guarded as they are potentially utilizable in producing nuclear weapons. They also remain radioactive for very long times; plutonium-239 has a decay half-life of 24,000 years.
The remaining 3% comprise products of U-235 or plutonium-239 fission, or daughter nuclei resulting from the subsequent radioactive decay of such fission products. These products range over a large number of elements and isotopes, but they include a number of isotopes that are radioactive with medium to very long half-lives. Among the most important long-lived radioactive remnant isotopes are: strontium-90 (half-life of 28.8 years); technetium-99 (211,000 years); iodine-129 (15.7 million years); and cesium-137 (30.1 years). Radioactive products with shorter half-lives, whose detection outside a containment vessel might signal radiation leaks, include iodine-131 (half-life of 8.0 days) and krypton-85 (10.8 years).
Spent fission fuel requires very specialized remote handling and storage. Ten years after removal from a working reactor, a spent fuel rod may still have a radiation level at its surface sufficient to deliver a lethal dose to humans in a few minutes. In some countries, spent fuel rods are sent to chemical reprocessing plants to extract the uranium and plutonium, which can then be reused in fuel for some types of fission reactors. The U.S. has not been reprocessing spent fuel rods and is encouraging other countries not to extract plutonium in this way, because of plutonium’s role in nuclear weapon proliferation. For example, India has used plutonium from reprocessing to develop their own nuclear weapons. Other countries, such as Pakistan, have relied on using highly enriched uranium to build nuclear weapons. Iran has built large underground facilities where they have used centrifuges to enrich the isotope U-235 in uranium. To the best of our knowledge Iran has not yet managed to produce nuclear weapons using this enriched uranium, but they are alleged to be close.
Because of the very long half-lives involved, the remaining waste must be disposed of permanently in very well-shielded sites that prevent any of the radioactivity from reaching the environment, including leaching into groundwater. The World Nuclear Association has a list of the many techniques that have been suggested to dispose of radioactive material so that the public is not harmed. Many of these techniques have been studied but never implemented. After a half-century of nuclear power, there is now international agreement that this high-level fission waste should be relocated to mined geologic repositories, but appropriate sites have been approved to date only in Finland and Sweden. In the meantime, spent fuel rods are usually stored temporarily on power plant sites. This temporary storage makes them vulnerable to exposure in the case of a catastrophic accident, such as the one at the Fukushima Daiichi power plant in Japan, which we will discuss in Section IV.
According to the Nuclear Regulatory Commission (NRC): “All U.S. nuclear power plants store spent nuclear fuel in ‘spent fuel pools.’ These pools are made of reinforced concrete several feet thick, with steel liners. The water is typically about 40 feet deep and serves both to shield the radiation and cool the [still thermally very hot] rods. As the pools near capacity, utilities move some of the older spent fuel into ‘dry cask’ storage. These casks are stainless steel canisters surrounded by concrete. Fuel is typically cooled at least five years in the pool before transfer to cask… At this time there are no facilities for permanent disposal of high-level waste. In the Nuclear Waste Policy Act of 1982, amended in 1987, Congress directed the Department of Energy to design and construct an underground geologic repository at Yucca Mountain, Nevada. DOE applied to the NRC for a construction authorization in 2008; however, DOE canceled the project in 2010 before the NRC completed its review.”
There are mainly political reasons why more suitable sites for disposal of radioactive wastes have not been successful in the U.S. The disposal of radioactive waste is a problem that will have to be addressed if countries continue to use nuclear reactors to generate power.
Nuclear meltdown:
Maintaining sufficient coolant flow is absolutely critical to prevent thermal runaway in a reactor core. Any accident, such as a failure of the water pump in the high-pressure circuit within the containment structure in Fig. III.3, or operator error that interrupts or constricts coolant flow, or fails to maintain the high coolant pressure in a PWR, risks a nuclear meltdown event, where the ongoing chain reaction continues to generate heat and the core temperature rises steadily, until it reaches the melting point of the uranium fuel itself. Operating fission power plants are required to have elaborate safety systems to prevent meltdowns. For example, in the event of a failure of some system elements, the control rods are programmed to drop all the way down, where they will shut off the fission process. Even then, however, the decay of radioactive nuclei built up in the fuel rods will continue to generate heat that must be transferred away from the reactor core. The containment structure is designed so that, in the event of a catastrophic failure of the system, whatever (non-nuclear) explosion might occur in the reactor from the buildup of hydrogen pressure and the radioactivity it might release would not escape the containment vessel. However, as we will see from historical accidents reviewed in Part II of this post, there are rare occurrences where the “fail-safe” procedures fail to prevent meltdowns.
The successive stages in a meltdown event are illustrated in Fig. III.4. If the core temperature surpasses the melting point of one or more fuel elements (the uranium oxide pellets themselves melt at 2,865°C), the fuel rod containers can be breached and radioactive isotopes can leach into the circulating coolant. If any appreciable portion of that radioactivity leaks outside the containment structure, people within surrounding areas of the power plant must be evacuated, quite possibly for long periods of time.

Once the fuel rods begin to melt, the non-circulating water surrounding them begins to boil away and steam pressure in the reactor core rises. In the worst cases, the vessel containing the reactor core may rupture, depositing highly radioactive material in the pit of the containment structure. Furthermore, at such extreme temperatures water molecules may dissociate, releasing potentially explosive hydrogen gas within the core. Despite multiple safety features intended to prevent such cooling losses, there have been a significant number of such core damage incidents in fission reactors worldwide. The three major worldwide reactor disasters that we discuss in Part II all involved at least partial meltdowns. And these disasters have strongly influenced public perceptions of fission power.
