July 9, 2021
Part II: The Blockbuster
Arthur Sackler died on May 26, 1987 of complications from heart disease. Because of his penchant for secrecy, combined with his dislike of being in hospital, he entered Presbyterian Hospital under a pseudonym, and did not notify his family, except for his third wife Jillian Sackler. By the time his extended family was notified, Arthur had already passed away.
Following his death, Arthur’s heirs had to unwind his financial dealings – in particular, his many businesses (at the time of his death, Arthur Sackler owned some 42 companies). In addition, Arthur had made numerous philanthropic commitments. Often, he had borrowed money to purchase art works that he then donated, and he had used corporate funds for some of his charitable donations. To make matters worse, a great many of Arthur’s commitments and business arrangements were basically handshake deals – Arthur was loath to put things in writing. A final straw was that in his will, Arthur Sackler left each of his four children by three wives $400,000. He also bequeathed to them the Medical Tribune newspaper business, which was worth roughly $30 million. However, he left his third wife, Jillian Sackler, the bulk of his $100 million fortune.
The various Sackler siblings, wives, and children made initial attempts to divide assets up in a relatively amicable fashion, but this rapidly devolved into a standoff where everyone involved “lawyered up.” There were arguments about the many properties owned by the Arthur Sackler families, and about the more valuable works of art in Arthur’s collection. But central to all these negotiations was the disposal of companies owned by the Sacklers.
During the legal wrangling over Arthur Sackler’s estate, Arthur’s first wife Else was asked if Arthur ever entered into “A business relationship with Mr. Frohlich.” “I don’t recall that he ever did,” she responded. The lawyers continued, asking if Else knew about “A proposal during Dr. Sackler’s lifetime” whereby stock in companies he created “would be sold and the proceeds donated to charity.” “Absolutely not,” replied Else Sackler. This was an extraordinary statement by Else, who as Arthur Sackler’s first wife had the most comprehensive knowledge of his business dealings, and who must have known of Arthur’s “multiple, intense, overlapping business relationships with Frohlich.”
Else Sackler’s statements denying Arthur’s business relationship with Frohlich and their ‘Musketeers’ agreement appear to have been bald-faced lies. In any case, these old agreements that the companies would pass on to the survivors and after all participants had died be sold off to charity were apparently forgotten. A crucial element of the disposal of Arthur Sackler’s estate then involved two of the Sackler companies, Napp Laboratories and Purdue Frederick. In 1987, Napp Laboratories were experiencing great success with the marketing of a time-release morphine pill called MS Contin in England, but the drug was brand-new in the U.S. And they had a new continuous-release drug that Purdue Frederick was hoping to market in the States. Mortimer and Raymond Sackler had no incentive to inform the heirs of Arthur Sackler of the tremendous potential of those two companies.
They were aided in this by Arthur Sackler’s own attorney, Michael Sonnenreich. In negotiations with Mortimer Sackler, Sonnenreich chimed in “I know what the value of companies are. This [Purdue Frederick] is a small company.” Following negotiations over dividing up his assets, Arthur Sackler’s heirs eventually sold their one-third share of Purdue Frederick to Mortimer and Raymond Sackler for $22 million. In light of what was to come, Patrick Keefe assesses this agreement as “a spectacularly foolish transaction.”
The widespread use of opiates in medicine at the end of the 20th century and beginning of the current century will be the subject of the remainder of this blog. Thus, at this time we will provide a very short review of the history of opiates.
A Brief History of Opiates:
Opiates are derivatives of a liquid extracted from the pod of the opium poppy. The qualities of opium – its ability to dull pain, to induce sleep, and its deadly and highly addictive properties – have been known for centuries. We know of the cultivation of opium poppies and uses for opium from Mesopotamia as far back as 3,400 B.C.E. Cultivation of opium subsequently spread from the Sumerians to Egypt, Persia and Greece. Even at that time, the properties of opium were known and appreciated. It is not certain to what degree the ancient people understood fully the addictive properties of opium.

Figure II.1: The Opium Poppy, the source of opioid drugs.
In 1803, Friedrich Sertuerner was the first person to isolate the active narcotic ingredient in opium. It was called morphine. Pure morphine is ten times as powerful as opium. For several decades, morphine was used as a painkiller in operations. For example, during the U.S. Civil War, morphine was widely utilized as a painkiller. As a result, it is estimated that as many as 400,000 soldiers became addicted. In the 19th century, opium and its derivatives appeared in a number of patent medicines, of which paregoric and laudanum were two examples. These products were recommended for all manner of conditions, including “malaise,” whatever that was. Also, many “soothing solutions” designed to quiet babies contained opium.

Figure II.2: An ad for Mrs. Winslow’s Soothing Solution, a 19th century patent medicine containing laudanum, a derivative of opium.
There was a concerted effort to find an alternative to morphine. The “Holy Grail” of these efforts was a drug that possessed the painkilling properties of morphine without any of its addictive potential. In 1874, Dr. Alder Wright synthesized diacetylmorphine; it was found that this was an extremely effective painkiller. In 1898, Bayer Laboratory chemist Heinrich Dreser also synthesized diacetylmorphine. Bayer subsequently patented the substance and called it heroin; the name derived from the German heroisch (heroic), since this is how workers who were tested with this product described their reaction.

Figure II.3: Glyco-Heroin, a patent medicine touted to relieve “cough.”
Initially, it was thought that heroin was not addictive. Thus, near the end of the 19th century, heroin appeared as an ingredient in a number of patent medicines. Bayer marketed heroin as a “safe” alternative to morphine, and they included it in cough syrup and for colds. Opiates also tend to make the user constipated, so medicines containing heroin were sold as antidiarrheal products. Doctors prescribed heroin for relief from menstrual cramps and respiratory problems. The result of this use was a dramatic surge in addictions in both the U.S. and Europe.
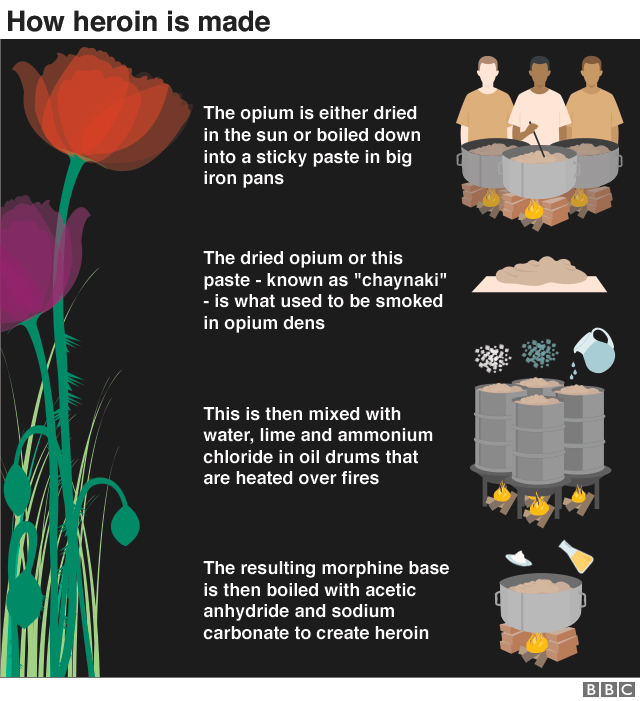
Figure II.4: A poster showing how heroin is produced from opium.
In 1908, President Theodore Roosevelt appointed Dr. Hamilton Wright as Opium Commissioner of the U.S. Wright issued the following statement regarding opium. “Of all the nations of the world, the United States consumes the most habit-forming drugs per capita. Opium, the most pernicious drug known to humanity, is surrounded, in this country, with far fewer safeguards than any other nation in Europe.” Wright’s statement was followed by a vigorous campaign to criminalize opium and its derivatives. In 1914, Congress passed the Harrison Narcotics Tax Act. This act placed severe controls on the use or prescription of products containing opium, heroin or cocaine.
Ten years later, Congress passed the Anti-Heroin Act of 1924. This made it illegal to make, import or sell heroin. The criminalization of these drugs was accompanied by a massive public-relations campaign. People who used heroin or other narcotics were called “dope fiends.” Addiction was described as a moral failing – people became addicts because they had loose morals and a severe weakness of character. Addiction was seen as the result of a self-abusing nature, so that addicts were blamed for their behavior that brought on this affliction. The other half of this coin was the evil “dope peddler,” who would try to hook users on drugs.
We now know that opiates have devastating neurological effects. A drug like heroin produces an incredible initial rush; it binds to opioid receptors in various areas of the brain. It is particularly absorbed in areas that govern pleasure and pain, and in areas that control sleep, heart rate and breathing. Continued use of opiates can actually “re-wire” the brain so that the user develops an acute craving for the drug. This leads to an addictive cycle: the user experiences a tremendous high when the drug contacts the brain. Then a feeling of release and lethargy ensues. But some time afterwards, the addict develops an overwhelming urge to repeat the cycle. Failure to score another hit can leave the addict nauseated, sweating and with tremendous anxiety. Addicts will do almost anything to acquire more drugs. In most cases, this leads to criminal activity in order to sustain a habit.
Purdue Frederick Markets MS Contin:
The pharmaceutical company Purdue Frederick was founded in 1892 by doctors John Purdue Gray and George Frederick Bingham. The name of the company was taken from the middle names of each founding partner. Purdue Frederick made a number of products, but their main sellers were a laxative called Senekot, a disinfectant Betadine and an earwax remover called Cerumenex. In 1952, the brothers Arthur, Mortimer and Raymond Sackler purchased Purdue Frederick.
Raymond Sackler was the head of Purdue Frederick, and he soon hired as assistant to the president his son Richard Sackler. Another company owned by the Sacklers, Napp Laboratories, developed a special time-release coating for pills. Pills with this coating would release the medicine slowly, with precise timing. Napp Laboratories had used this technique, which they called Continus, on a drug for asthma patients.
Raymond Sackler’s brother Mortimer was in charge of the Sackler family’s overseas operations. When Napp developed a time-release morphine pill called MS Contin, Purdue Frederick produced and marketed it in the U.K., releasing it in 1980. MS Contin became a revolutionary drug in Britain. The time-release aspect meant that cancer patients no longer needed to enter a hospital every time they needed morphine for their pain. In the hospital, morphine was delivered in a drip, where the powerful drug was absorbed very slowly. But with the time-release pill, the morphine could be released just as slowly as with a drip, so the patients could take the pills at home or in a hospice.
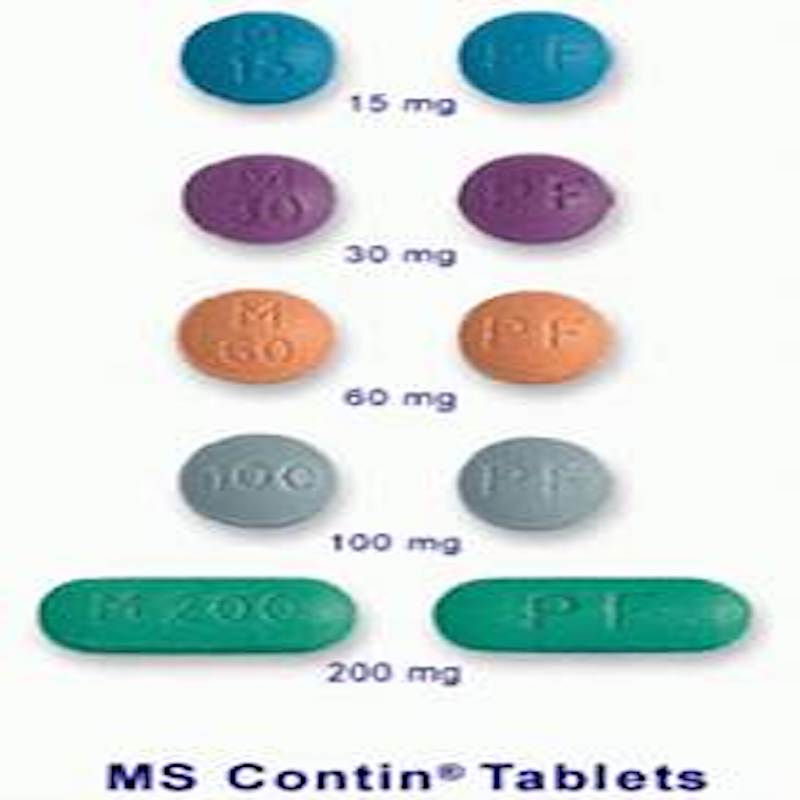
Figure II.5: A range of doses of morphine available in MS Contin tablets.
MS Contin received lavish praise in some quarters. One physician was quoted in the London Times describing MS Contin as among “the most important advances in narcotics this century.” Another doctor called it “The most important step forward in drug control of pain since morphine itself.” So Purdue Frederick was determined to market MS Contin in the U.S. as well. However, this was likely to be an extremely time-consuming process. The Food and Drug Administration (FDA) required a lengthy and rigorous process before approving a new drug for release in the U.S. Furthermore, the process of approving a drug containing an opiate such as morphine (a so-called Schedule II narcotic) would be even more deliberate.
The Sackler family had witnessed the many ways that their patriarch Arthur had revolutionized the field of medical advertising. Arthur Sackler had devised a series of principles that we will call the “Arthur Sackler playbook.” His extended family would adhere to these principles in their marketing of MS Contin and later OxyContin. These principles included:
- Secrecy: Arthur Sackler went to great lengths to keep his name out of the papers (with respect to his business ventures). He never gave interviews about his businesses (he made an exception for his philanthropic ventures), and he went to considerable lengths to keep his name off the lists of many companies that he owned.
- Expediency: Sackler dealt with ethical issues such as conflict of interest by simply going around them. It would have been a conflict of interest for his McAdams medical advertising firm to represent different companies making competing products. So Arthur set up his friend Bill Frohlich in business as a competing agency; and he concealed the fact that Arthur was a silent partner in Frohlich’s firm. Also, Arthur had a secret interest in the medical data collecting firm IMS.
- Hyperbole: One of Arthur Sackler’s advertising campaigns included “testimonials” for a product from various physicians. When people tried to contact those physicians, they discovered that they did not exist. Arthur’s ad campaigns for Valium contained many claims about the product that were later found to be based on little or no evidence.
- Ignoring or Denying Side Effects of Medicines: Arthur Sackler played down the side effects of Librium and Valium; he continued to tout those products as safe even after side effects were noted.
- Use of Sales Representatives: Sales representatives had been used before Arthur Sackler, but they tended to be small in number. Arthur’s sales campaigns expanded the number of sales reps tremendously. Also, sales reps provided doctors with free meals and “swag” – materials branded with the name of the med, or the name of the company.
- Saturation Advertising: Arthur Sackler greatly increased the amount of advertising to physicians. He would produce multi-page inserts in publications such as the New England Journal of Medicine. He bombarded doctors with lavishly-produced brochures that were used by the physicians or handed out to patients.
In an example of “expediency,” Purdue Frederick took a brazen step with MS Contin. They argued that although the time-release mechanism was new, the drug was simply morphine, which had already been approved for use in the U.S. They claimed that this allowed them to put the drug on the market without FDA approval! Purdue knew that they were skirting the law – at that moment, a federal regulation was being introduced that would forbid the release of “new formulations” of approved drugs, and require them to go through the FDA’s New Drug Application. So Purdue Frederick’s head lawyer Howard Udell urged the company, “Before this [new regulation] goes into effect, let’s make MS Contin – and put it on the market.”
Purdue Frederick then went into production of MS Contin, releasing the drug in November 1984. Purdue sent around teams of sales representatives to doctors and hospitals pushing their new drug. They marketed MS Contin without requesting FDA permission; moreover, they did not even inform FDA that they were releasing it in the U.S.!
Three months after the release of MS Contin, the FDA informed Purdue Frederick that they could not market their drug without completing the FDA’s New Drug Application and obtaining permission for the product. In theory, this meant that Purdue would have to pull MS Contin off the market, submit the New Drug Application, and wait for years before the drug was approved for use. Instead, the Purdue lawyers descended on Washington to plead their case before the FDA. They were armed with testimonials from cancer doctors and patients, who voiced fears that they would suffer if MS Contin was pulled from the market.
At the same time, the company attempted to garner political support for their tactics. Purdue Frederick argued that this was simply a misunderstanding, and that they never realized their drug would need FDA approval (despite Howard Udell’s memo that showed they were keenly aware of their obligation). Purdue lawyers also solicited support from the Reagan Administration (the Sacklers were significant donors to Republicans). The net result was that the FDA backed down.
Instead of requiring that Purdue Frederick take MS Contin off the market, the FDA allowed them to continue to market the drug, while they prepared the New Drug Application and went through the FDA approval process. This allowed the company to continue marketing and selling MS Contin, which eventually became the biggest seller ever for Purdue Frederick; annual sales eventually reached $400 million. Despite MS Contin’s success the company, and in particular its president, Richard Sackler, had visions of much larger sales for an opiate product. But for that, they would need the support of a group of physicians and researchers. Most fortunately for them, a new “pain management” paradigm was being proposed. Purdue would work hand-in-glove with advocates for a revolutionary new approach to pain management.
The “Pain-Management” Lobby:
The continuous-release morphine tablet MS Contin was generally approved for cancer patients who were in the final stages of life. As a Schedule II drug, the administration of morphine was permitted, under carefully controlled conditions, to provide these terminal patients with relief from pain at the end of their life. The revolutionary aspect of MS Contin was that patients could take it at home, without having to enter a hospital every time they needed pain relief.
But Purdue Frederick had a vision where opiates could be prescribed to a much larger segment of the population. At just this time, a group of physicians were calling for a new approach to pain management. Purdue Frederick and the Sacklers realized that they could exploit this revolutionary approach to pain, in order to expand their production and sales of opiates. They therefore used all the elements from the medical-advertising playbook established by Arthur Sackler. This included the following steps:
- A campaign was orchestrated in order to change the way the medical profession dealt with pain. Previously, pain was considered a symptom to be alleviated with medical and surgical methods. However, a group of physicians began to argue that “pain” should be considered its own medical condition, and that “pain management” itself should be a specialized field of medicine. There were physicians such as John Bonica who claimed that America was suffering from an “epidemic of pain,” where people suffering from excruciating chronic pain were not receiving the treatment they needed.
Another advocate of this “new pain management” paradigm was Russell Portenoy, who was the founding head of the Department of Pain Medicine and Palliative Care at Beth Israel Medical Center. Another was Dr. Kathleen Foley, Foley and Portenoy were co-authors of a seminal paper that explored the sustained use of opioids for pain relief. Portenoy himself described opioids as “a gift from nature.” And what a gift they would turn out to be.

Figure II.6: Dr. Russell Portenoy. His strong advocacy for using opiates to treat chronic pain gave him the nickname ‘King of Pain.’
- The new pain management doctors argued that pain should be considered a “fifth vital sign.” That is, in addition to the four vital signs (pulse, blood pressure, body temperature, and respiration), doctors should consider pain as an additional vital sign. In fact, the American Pain Society trademarked the slogan “Pain: the Fifth Vital Sign.” The new campaign paid off. In 1998 the Veterans Health Administration declared that pain was a fifth vital sign. The same step was taken by the Joint Commission for Accreditation of Healthcare Organizations (JCAHO), the group that accredits 16,000 healthcare organizations across the country. This was somewhat problematic, because unlike the other four vital signs, there is no precise measure of pain; one still largely relied on patient self-description of the severity of pain on a subjective “1 to 10” scale.
- Purdue Frederick seized upon these ideas. Together with other pharmaceutical companies that produced pain meds, Purdue Frederick underwrote the formation of “astroturf” groups such as the American Pain Society and the American Pain Foundation. While these appeared to be independent groups of physicians and researchers, they received their financial support from Big Pharma manufacturers of pain meds. In 1984, Raymond Sackler’s son Richard Sackler helped organize the International Symposium on Pain Control. Speakers at this conference focused on changing “worldwide concepts of pain theory and management.”
Many of the speakers at this conference referred to their use of MS Contin in alleviating pain. They attested that in their view, morphine was an effective way of alleviating pain. They wanted to remove the “stigma” attached to morphine. As Richard Sackler noted, this stigma arose “because of a popular understanding shared by both professionals and laymen that morphine was an end-of-life drug.” Furthermore, they needed to counteract the widely-held view that morphine was addictive. Numerous speakers at this conference disagreed with this perception. For example, Dr. Eckhard Beubler asserted “Addiction does not occur in patients requiring morphine for pain control.” Radiation oncologist Jerome Ramagosa claimed that addiction was a “psychological malady” that only occurred when it was abused by “those who do not need it.” A group of doctors attending this conference issued a joint statement on pain management. They stated, “Morphine is the safest and best drug for the control of severe chronic pain.”
- Those in the “pain management” movement repeated Eckhard Buehler’s claim that addiction was almost unknown among patients receiving morphine or opiates from a physician. Part of this was related to the stereotype of drug users in the U.S., that we discussed in our review of the history of opiates and their uses. After heroin was made illegal in 1914, there was a major national campaign to portray illegal drug users as morally deficient. “Dope fiends” were described as people with loose morals and poor self-control, and they were blamed for their addiction. This created a vicious cycle: pain management advocates claimed that addiction was extremely rare in patients receiving opiates under a doctor’s care. The only cases of addiction would be through people who had “addictive personalities,” and who would “abuse” the drug. Thus, the pain management lobby attempted to escape any liability for addiction arising from treatments with opiates, since only those patients who were “prone” to addiction were abusing the drugs. To the best of our knowledge, the Sackler family still maintains that there was nothing wrong with the drug OxyContin, or with their sale of the drug, and that the only problem was that some irresponsible people abused the drug.
Portenoy’s Complaint: Addiction Is Less Than 1%:
In 1991, Purdue Frederick spun off the company Purdue Pharma to continue research and marketing of new pain meds. The “pain management” community, funded and spurred on by Purdue, would frequently stress that, when opiates were prescribed by a physician, addiction was extremely uncommon. This claim was so widely made by the pain management researchers and repeated by Purdue Pharma, that one would assume it relied on rigorous and exhaustive studies. Such studies would be particularly crucial, in light of the well-known highly addictive properties of opiates. In fact, a major source for this claim came from a single work by Jane Porter and Herschel Jick. Published in the New England Journal of Medicine in January, 1980, the title of this submission was Addiction Rare in Patients Treated With Narcotics.

Figure II.7: The ‘paper’ by Porter and Jick, in its entirety (101 words). It was cited over 600 times as a ‘landmark study’ showing that patients taking opioids would not develop addiction. It was nothing of the kind.
This paper was eventually cited more than 600 times (the average citation count for a paper in that month’s journal was 11). It became a mantra for the new breed of pain specialists – “Addiction is rare in patients treated with narcotics – the proof? The paper by Porter and Jick.” The paper was so widely quoted that a 1990 Scientific American paper referred to it as an “extensive study,” and a 2001 story in Time magazine called it a “landmark study.”
But what exactly was this Porter & Jick “landmark study”? Was it a double-blind study with extensive statistics that compared outcomes for patients treated with and without opioids? Was it a comprehensive investigation of the possibility of habituation or addiction for patients taking opiates? Did it follow a set of patients over a long period of time, and contrast the outcomes of patients treated with and without opiates? Had it undergone rigorous review by experts in the field? Was it even a paper? The answer to all these questions is “No.” The Porter & Jick “paper” was actually a letter to the editor; it consisted of 101 words, as shown in Fig. II.7. It was not peer-reviewed, and it was only 5 sentences in length. The letter referred to patients who were treated in a hospital; they were given extremely small doses of opiates under careful supervision; also, Porter & Jick did not follow the patients after they were released from the hospital.
So, the claim that “Porter & Jick proved that addiction is rare for any patients treated with narcotics” is totally unfounded. But it did not stop there. The “pain management” lobby further strengthened their claim to state that “For patients treated by doctors with opiates, the rate of addiction is extremely small, in fact less than 1%.” As we will see, the OxyContin sales reps sent around by Purdue Pharma to doctors across the country, many of them primary-care physicians with no background in pain management, repeated this “addiction less than 1%” claim like a mantra.
There were two other “major studies” that were touted by pain advocates such as Russell Portenoy. One of those was a 1977 study of migraine sufferers called Drug Dependency in Patients with Chronic Headaches published in the journal Headache. Portenoy claimed that the migraine study found only “three problem cases among 2,369 patients.” However, the three “problem cases” were actually found in a much smaller group of 62 patients who were in a separate category “because they had taken either painkillers or a combination of painkillers and barbiturates for at least six months before coming to the [headache] clinic.” And Portenoy did not quote the conclusion of the migraine study, which specifically warned against using narcotics for headache sufferers: “There is a danger of dependency and abuse in patients with chronic headaches.”
A third article had appeared in 1982 in the journal Pain; it reviewed the history of patients who were given narcotics while undergoing debridement (this is an extremely painful process where burn patients have dead skin removed from their bodies, in order to provide more rapid healing). Portenoy also claimed that these burn victims had shown no tendency towards addiction or abuse. However, like the other two studies, this study did not follow the patients for long periods of time, hence no conclusions could be drawn about the effects of long-term use of opiates.
And the hyperbole did not stop there. Pain management advocates then claimed that there was no upper limit to the amount of opiates prescribed. Since addiction was exceedingly rare when opiates were prescribed by physicians, then when patients complained that their dosage of opiates did not last for the specified 12 hours, or that their dosage was now failing to alleviate their pain, the solution was – give them larger doses of opiates (or opiates more frequently)! This was a recipe for disaster. As was well known, a person taking opiates builds up a tolerance to the drug. The amount of opiate needed to provide the same outcome keeps increasing. So the practice of prescribing ever greater amounts of opiates could have been predicted: this practice would lead to a great many users becoming habituated to the drug, and eventually becoming addicted.
But the pain management advocates (one could even call them the “opiate pushers”) continued on undeterred. In fact, they rationalized their behavior in a number of ways. One was to assert that “A person in pain would not experience the euphoria associated with opioids, because the pain will mask the euphoria.” As far as one knows, this was simply a myth. A second tactic was to blame the addict. Since “addiction was extremely rare” in patients treated with opiates by doctors, then the only people who became addicted must be “dope fiends.” They are either abusing the drug, or they had “addictive personalities.” Thus, the fault was not with the opiates, nor Purdue Pharma that marketed the drugs, or the doctors who prescribed them – the fault was with the addicts, who were deviants with loose morals and abusive tendencies. This was a continuation of our national mythology about drugs and addiction, a by-product of the decades-long “war on drugs.”
Russell Portenoy was one of the most outspoken advocates of the “new pain management” philosophy. He claimed that the American medical community had “sharply underestimated the benefits of opioids and sharply overestimated the risks.” Portenoy doubled down on this assertion: he claimed that Americans had an irrational fear of opioids, which he called “opiophobia.” Portenoy claimed that in cases where patients treated with opiates by physicians became addicted, there were often “predisposing psychological, social and physiological factors.” That is, some people just have addictive personalities and that’s why they abuse opiates. Note that this attitude can be used as a “get out of jail free” card for the pain management advocates, pharmaceutical companies that manufactured and distributed opiates, and doctors who prescribed them: since the only people who developed an addiction to their products had “pre-existing addictive conditions,” this would free those producing or marketing the products from any liability for their actions.
The pain-management advocates turned out to be great at convincing the public to aggressively treat pain with narcotics. They revolutionized the treatment of pain and created the new field of pain management. Now, doctors were no longer hesitant to prescribe opiates for pain – quite the reverse, the medical community was now told that doctors could be sued if they failed to prescribe opiates for chronic pain! But the pain-management mavens were lousy researchers. They all accepted the Porter & Jick “study” as gospel, without ever reviewing that letter with a skeptical eye. Also, they failed to consult with their colleagues. Pain-management advocates like Kathleen Foley and Russell Portenoy were apparently not working closely with specialists in addiction, who could have pointed out the severe weaknesses in their assumptions. They simply barged ahead, accepting the consulting fees from “astroturf” foundations funded by the pharmaceutical industry, and spreading the gospel of opioid pain management throughout the medical community.
The “pain management” advocates also focused their attention on prescription monitoring regulations. In the 1990s, about 14 states had databases that tracked the dispensing of all restricted drugs, such as Schedule II drugs that included morphine and oxycodone. Such systems could alert authorities when physicians began writing prescriptions for unusually large amounts of controlled substances. Dr. David Joranson, head of the Pain and Policy Studies Group at the University of Wisconsin, was an outspoken critic of prescription-monitoring systems. In a 1993 paper in The American Pain Society Bulletin, Joranson wrote “Requiring the use of these prescription forms would send an unmistakable message to physicians that prescribing controlled substances could give them an unwarranted profile with the police or licensing authorities if they order more than a minimal amount for patients in pain.”
So Joranson wanted to shut down one of the potentially most useful programs to combat abuse of dangerous opioids, in order to allow doctors to prescribe virtually unlimited amounts of Schedule II narcotics to their patients. As we will see, these “pain management” advocates helped allow an explosion of “pill mills” in the U.S. In my own home town of Bloomington, Indiana, we had at least two pill mills. In Dec. 2012, anesthesiologist Kamal Tiwari pleaded guilty to defrauding insurance providers and prescribing narcotic drugs to addicts. The Federal charge claimed that Tiwari “dangerously over-prescribed controlled substances to known drug addicts, contributing to the overdose deaths of multiple patients.” Tiwari served three years in prison.
In Dec. 2017, Dr. Tristan Stronger pleaded guilty to five charges including issuing an invalid prescription and insurance fraud; in exchange for his guilty plea, 50 additional charges were dropped. Stronger owned pain clinics in Peru, Indianapolis and Bloomington, Indiana. Investigators said Stronger saw as many as 100 patients in a single day, and traded pain pills for work on his farm. He was sentenced to 10 years of probation but served no jail time.
Purdue Develops OxyContin:
The drug MS Contin had proved to be a big seller for Purdue. Sales of the drug, mainly as a time-release morphine pill for cancer patients, were approaching $400 million per year. But the company wanted to expand their reach, so they produced a time-release pill containing the chemical oxycodone. Oxycodone was already an ingredient in prescription drugs such as Percodan and Percocet. A Percodan tablet contained 4.8 mg of oxycodone hydrochloride and 325 mg of aspirin, while one type of Percocet tablet contained 5 mg of oxycodone hydrochloride and 325 mg of acetaminophen.

Figure II.8: The Stamford, Connecticut headquarters of Purdue Pharma, the company that developed and marketed OxyContin.
Because oxycodone was included (at very low levels) in existing prescription drugs such as Percodan or Percocet, the public perception was that oxycodone was not as strong as morphine, and this perception was shared by many physicians. In fact, just the opposite was true: oxycodone is roughly twice as potent as morphine. But Purdue Pharma went to considerable lengths to prevent doctors from becoming aware of the actual potency of oxycodone. Because of the strength of this chemical, it would require very strong safeguards to prevent users from becoming dependent on oxycodone.
It is not clear who at Purdue first suggested that the company develop a timed-release formula using oxycodone instead of the morphine in MS Contin. Some attribute this idea to Kathe Sackler, the daughter of Mortimer Sackler and a member of the board of Purdue Pharma. Others say that Purdue staff doctor Robert Kaiko first suggested using oxycodone. In any case, Purdue Pharma and their new head of marketing, Michael Friedman, were enthusiastic about the idea. And, in a memo marked “Very Confidential” to Raymond, Mortimer, and Raymond’s son Richard Sackler, Friedman raised the idea that OxyContin (as the drug was eventually named) might be marketed to a much broader segment of the American public. Instead of the drug being restricted just to cancer patients, Michael Friedman suggested that the company should attempt to “expand the use of OxyContin beyond Cancer patients to chronic non-malignant pain.” In that case, OxyContin might be offered to people suffering from any form of chronic pain: “back pain, neck pain, arthritis, fibromyalgia,” etc. Friedman then suggested that “We establish a literature” to support such an argument. Such a literature would then recommend OxyContin “for the broadest range of use.”
Marketing a drug as potent as oxycodone to the general public, for a wide variety of chronic pain conditions, would essentially reverse decades of experience with opiates. The history of addiction from opium, morphine, heroin and other opiates had resulted in strict controls on using these and other Schedule II drugs. In order to obtain approval for such widespread marketing, it would be necessary to convince doctors and lawmakers that these strict controls were unnecessary with oxycodone. Fortunately for Purdue Pharma, their desires dovetailed with a new effort to remove the stigma from opiate use. Physicians and researchers in the new “pain management” movement were eager to allow the use of such powerful drugs. And Purdue Pharma was only too happy to assist them.
So Purdue Pharma developed a time-release drug containing oxycodone, called OxyContin. Because oxycodone was less well known than morphine, it did not suffer from the same stigma that was attached to morphine, namely that it should be taken only by patients in the terminal stages of a disease such as cancer. The American public had also been softened up by the paper by pain-management advocates Portenoy and Foley, who had proposed the use of opioids for treating chronic pain. They foresaw “The possibility of long-term pain relief from opioid therapy, without the development of … serious adverse effects , including drug abuse.” As Patrick Radden Keefe notes, “The evidence [behind this study] was mainly anecdotal.”
The FDA Approves OxyContin:
Purdue Pharma submitted OxyContin to the FDA for its approval as a scheduled narcotic. Normally, Purdue would have been required to carry out extensive testing to ensure that the drug could not lead to abuse or addiction. However, Purdue Pharma claimed that their time-release formula made it impossible to be abused. Addiction, they argued, was based on cycles of peaks and troughs, and the continuous time-release of oxycodone meant that addiction was not possible. So Purdue Pharma did no research on habit-forming aspects of their drug.
The application for FDA approval had to be approved by the head of pain medication at that agency, Curtis Wright. Initially, Purdue Pharma was requesting FDA approval for a drug that would be provided to cancer patients, just as MS Contin had been. Purdue proposed a “package insert,” the booklet of information that is included with every bottle of medicine. When Wright reviewed the Purdue insert, he remarked that “he’d never seen an insert that contained so much promotional and marketing material.” Wright said that Purdue would have to remove the extraneous material; however, all that material was included in the OxyContin package insert.

Figure II.9: Dr. Curtis Wright. He was the FDA official personally responsible for approving OxyContin. The ease with which OxyContin could be abused was a significant factor in the development of the opioid epidemic. After Wright approved OxyContin, he resigned from the FDA; one year later, he was hired by Purdue Pharma.
During the FDA approval process, Purdue Pharma assiduously courted Wright. Eventually, Wright took the highly unusual step of informally contacting various staff at Purdue to discuss their application. In fact, on occasion “he instructed Purdue to send him certain materials at his home office, rather than at the FDA.” Eventually, Wright appeared to be acting more like an advocate for Purdue’s drug than the administrator charged with approving the application. A particularly striking feature was a sentence in the final package insert: “Delayed absorption, as provided by OxyContin tablets, is believed to reduce the abuse liability of the drug.” The statement is remarkable because it is presented with no references to corroborate it – and as we now know, the only “proof” of that claim was the Porter-Jick letter, which did not prove this at all. In fact, one of Curtis Wright’s colleagues at the FDA, Diane Schnitzler, wrote to Wright that this statement “Sounds like B.S. to me. Any truth to this statement or legit reason to be in label?” In Barry Meier’s 2003 book Pain Killer, he states that in an FDA report, Wright stated that “Care should be taken to limit competitive promotion” of OxyContin.
The FDA approved OxyContin on Dec. 28, 1995. Purdue president Richard Sackler told his staff, “This didn’t just ‘happen.’ It was a deftly coordinated, planned event.” Indeed, it was coordinated and planned more carefully than anyone realized at the time. Shortly after the approval, Curtis Wright resigned from the FDA. He joined a small pharmaceutical firm, but after one year he went to work for Purdue Pharma. Later, Wright would defend his hiring by Purdue; it had nothing to do with his favoritism towards that company, but was because he had been “a particularly fair and effective FDA reviewer.” However, Patrick Radden Keefe reports that even before leaving the FDA, Wright had contacted Purdue Pharma about the possibility of a job. It was realized that it might look bad if Wright joined Purdue Pharma immediately after approving their drug OxyContin. So they agreed that Wright should wait a year, a cooling-off period, before approaching Purdue Pharma for a job.
Curtis Wright took advantage of the well-known “revolving door,” whereby staffers in a government agency proceed to work for the very companies whose products they had been regulating. As we have shown, OxyContin had been approved without any rigorous studies being carried out to check on the addictive properties of the drug. And despite the fact that OxyContin was a Schedule II narcotic, Purdue Pharma’s application was approved in record time. Purdue Pharma, of course, was jubilant about their success. Despite the fact that the drug had been discussed as a remedy for cancer patients, once they had FDA approval Purdue could market it to anyone. And the pain-management advocates were also celebrating the release of opiate drugs. In 1996, the year that OxyContin appeared on the market, Dr. Kathleen Foley said “We went from not having very potent drugs … to an oral morphine form and to a pharmaceutical industry that wanted to promote it. So we, for the first time, then, had a marketer and a distributor for a drug that we had, and an educator. We knew then that oral morphine was effective. We can get away from these silly elixirs and cocktails into tablets that people can take once or twice a day, and we’re into a revolutionary field of pain management … It was the drug-delivery device that changed, not the drug, and with that the whole mentality, ‘Well, now that we have this drug, we can treat pain.’ Really extraordinary.”
Really extraordinary, indeed. We will see just how revolutionary OxyContin was.
OxyContin Is Marketed: A Juggernaut Ensues
In January 1996, Purdue Pharma released the pain drug OxyContin on the market. Richard Sackler, president of Purdue Pharma, was jubilant over the fact that OxyContin had been approved by the FDA in less than a year. He predicted great things for their new drug. “The launch of OxyContin tablets will be followed by a blizzard of prescriptions that will bury the competition,” said Sackler at the launch event for OxyContin (the “blizzard” metaphor referred to a major blizzard that had just struck the East Coast). “OxyContin tablets is the most important product launch in the company’s history. In the years to come, we will look back on this week as the beginning of a new era for our business, and for ourselves … There is absolutely nothing that is ethical and legal that we won’t do to make the greatest sales force on earth even more successful!”
Arthur Sackler had earlier used teams of sales reps to “sell” doctors on Roche’s tranquilizer Librium. Now, Purdue Pharma would repeat this by unleashing their sales reps on doctors across the country. But before they went out on the road, the sales reps were carefully prepped to sell OxyContin to doctors, and they were drilled on arguments to overcome any resistance the physicians might have to prescribing opiates to their patients. The sales reps would strongly advocate that doctors switch their patients from any prior pain meds to OxyContin. OxyContin, said the sales reps, “Is the drug to start with and to stay with.” This product was advertised for treating anything “from moderate pain to severe pain.” A patient could take it for chronic back pain, or knee pain, or fibromyalgia, or cancer pain. According to the sales reps, patients could take it for short periods, or, if necessary, they could continue to take it indefinitely.
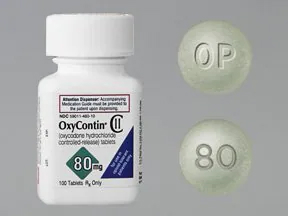
Figure II.10: OxyContin tablets containing 80 mg of pure oxycodone, inside a coating that would release the drug over time.
Doctors would quite likely inquire about the possibility of abuse or addiction: after all, OxyContin was an opiate. The sales reps were taught to first quote from the package insert: “The delivery system is believed to reduce the abuse liability of the drug.” There it was: FDA approved! The reps also stressed that the time-release method insured that the “peaks and troughs” associated with opiate abuse would be absent. If doctors persisted in questioning the safety of OxyContin, the sales reps would respond by quoting the medical literature. “A survey of more than 11,000 opioid-using patients, taken over several years, found only four cases of documented addiction,” they were trained to say, despite the fact that this was seriously misleading.
As we have pointed out, the sales reps were referring to the “Porter & Jick paper;” remember that this was neither a peer-reviewed paper nor a long-term study, but a 5-sentence letter in the New England Journal of Medicine. The patients in question were being given small doses of opiates in a hospital setting, and the patients were not followed after they were released from hospital. Another of the sales reps selling points was taken from the pain-med advocates: “Addiction in patients given opiates by physicians is extremely rare, less than 1%.” Again, this was a claim made with no large-statistics studies to back it up.
But the sales reps were extremely persuasive. Sales of OxyContin took off. And, to be sure, OxyContin gave rapid and notable relief from pain – after all, it was a powerful opiate. OxyContin tablets were issued in exceptionally large doses – there were 10, 20, 40 and 80 mg tablets (by comparison, the drug Percodan contained less than 5 mg of oxycodone along with 325 mg of aspirin; apart from the time-release coating, OxyContin was pure oxycodone).
In addition to the phalanx of sales reps, Purdue Pharma sponsored “pain management seminars” that ostensibly informed physicians about the latest developments in this field. “Purdue established a ’speakers bureau’ of pain management specialists who participated in” these events, which were often held at resorts such as Scottsdale, Arizona or Boca Raton, Florida. Doctors were paid to attend the seminars, where they were bombarded with OxyContin swag, and talks by physicians consulting for Purdue Pharma. Patrick Radden Keefe reports that in the first five years after OxyContin was released, Purdue sponsored seven thousand pain seminars (a number that is hard to believe).
Purdue Pharma poured in resources to increase sales of OxyContin, and simultaneously to remove the stigma over prescribing opiates for non-malignant chronic pain. They paid their sales reps handsomely when they succeeded in their sales quotas. Most pharmaceutical companies set limits on the size of bonuses they would pay to their sales reps, but Purdue Pharma would allow their reps to earn unlimited bonuses. The company also allocated over $9 million in food for doctors, either at the seminars or from individual wining & dining by Purdue sales reps. Furthermore, while most pharma companies paid bonuses for the number of prescriptions made by the doctors they visited, Purdue’s bonuses were based on the dollar value of the business; thus Purdue sales reps had a strong incentive to pressure their doctors to prescribe the maximum dose of OxyContin.
Richard Sackler was keenly aware of the benefits of this blanket coverage for his product. In 1996, Sackler wrote in an e-mail to marketing director Michael Friedman that “physicians who attended the dinner programs or the weekend meetings wrote more than double the Rxs [prescriptions] for OxyContin compared to the control group.” Purdue Pharma kept detailed records on those physicians who prescribed large amounts of OxyContin; apparently the company referred to those doctors as “whales,” and sales reps explicitly targeted those individuals. In addition, Purdue concentrated their sales efforts on “places where there were a lot of family physicians, where people had workers’ comp, injuries they had sustained on the job, disabilities.” The company also targeted family physicians who were “opioid naïve” – their only understanding of opiate issues would come from Purdue’s own sales reps and company brochures. By the year 2003, “more than half of the prescribers of OxyContin nationwide were primary-care doctors, who had little pain-management training and were under pressure to get patients in and out of their offices. Oxy prescriptions for chronic pain rose from 670,000 in 1997 to 6.2 million in 2002.”
As we have noted, OxyContin turned out to be a powerful painkiller. The company began to accumulate testimonials from people who had been living with chronic pain, and for whom the drug gave immediate relief. In the first year following its release, OxyContin sales amounted to $44 million; that amount doubled in the second year, and it doubled again in the third year. Purdue also wanted to issue free samples of OxyContin; however, it was prohibited to issue free samples of Schedule II drugs. They got around this prohibition by issuing coupon cards to doctors. Physicians would give the coupons to patients to cash in at a pharmacy, where they would obtain a free 30-day prescription. This giveaway bore a creepy similarity to the actions of illegal drug dealers, who often offer free “first hits” as a means of getting people hooked on their product. Purdue Pharma issued 34,000 free prescriptions before they suspended that program in 2001.
At the Purdue Pharma annual meeting in 2000, Richard Sackler told his employees that “You are part of a legend in the making.” Sales of OxyContin “have outperformed our expectations, market research, and fondest dreams.” The drug was on track to become one of the biggest sellers in pharmaceutical history. In the first four years of its release, OxyContin surpassed $1 billion in sales; this pushed it past another legendary drug, Viagra. Apparently the only obstacle to OxyContin sales was that the company could not manufacture the drug sufficiently fast to keep up with the demand.
Purdue Pharma introduced a new incentive program called “Toppers.” It rewarded those sales reps who produced the largest OxyContin sales. Prizes might be all-expenses-paid trips to Caribbean locales. Eventually, the bonuses alone paid to sales reps topped $250 million per year.
The Sackler family reveled in the success of their new oxycodone drug. Richard Sackler, the president of Purdue Pharma, was especially pleased with the sales of the drug and the success of their sales representatives. What could possibly go wrong?
