July 9, 2021
Part III, Purdue’s Reign of Death:
Unfortunately, the use of OxyContin was causing problems; and those problems should have been readily obvious to Purdue Pharma. They were, after all, marketing an opiate. The first issue that manifested itself was that the drug was marketed as relieving pain for 12 hours. The recommended dose was two pills per day; but many users found that OxyContin failed to relieve their pain for that long. Purdue Pharma knew about this, as this issue had surfaced in studies made before obtaining FDA approval for OxyContin. In one study, researchers prescribed the drug to ninety Puerto Rican women following surgery. “More than a third of the women given OxyContin complained about pain in the first eight hours, and roughly half of the women ended up requiring more medication before the twelve-hour mark.”
Purdue’s application to the FDA for drug approval claimed that OxyContin provided 12 hour relief “in approximately 90% of patients.” In addition to the Puerto Rican project, in one study of 164 cancer patients given OxyContin, one third of the patients withdrew from the study because the pain treatment was “ineffective.” The researchers then altered the study to allow patients to take supplemental meds in between the 12-hour doses of OxyContin. In another study of 87 cancer patients, 95% of the participants resorted to supplemental meds, so-called “rescue medications” at some point in the study.
If patients required more pain meds before the 12-hour period, Purdue Pharma’s solution, and the advice offered to physicians by sales reps, was to move to an OxyContin pill that contained more oxycodone. After all, their mantra was that “addiction is almost unheard-of in patients prescribed opiates by physicians.” So Purdue Pharma recommended that doctors prescribe more powerful doses of OxyContin. This was also in line with another unsubstantiated belief that was shared by Purdue Pharma and the “pain management” advocates: when administered under a doctor’s care, there was almost no upper limit on the strength of opiates that could be administered. This was a remarkable assertion, as it contradicted virtually everything we had learned from studies of addiction.
Purdue Pharma emphasized the claim that OxyContin would provide 12 hours of pain relief. This was a major factor in its desirability, as competing drugs such as Percodan or Percocet had to be taken every six hours. So the ability to take one drug at, say, 8 am and a second at 8 pm, and have all of your pain relieved, was a major selling point and one that Purdue stressed in its advertising campaign for its drug. Purdue stated that one Oxy in the morning and one before bed would provide “smooth and sustained pain control all day and all night.”

Figure III.1: A Purdue Pharma sales ad for OxyContin, stressing that each pill provides 12 hours relief from pain. It shows two cups for pills, one with the label “8 am” and the second with the label “8 pm.”
However, as the Los Angeles Times reported, because of OxyContin’s similarity to heroin, when the drug doesn’t relieve pain for 12 hours, “patients can experience excruciating symptoms of withdrawal, including an intense craving for the drug.” But Purdue steadfastly stuck to its claim that OxyContin gave patients 12 hours of pain relief. In fact, both Purdue Pharma’s market dominance and the high cost of OxyContin (a bottle of 80-mg Oxy tablets cost $630) were dependent on the unique 12-hour feature. Without that advantage, painkillers like generic morphine could be prescribed at pennies per pill. And data obtained by Purdue Pharma in 2000 showed that “one in every five OxyContin prescriptions was for use every eight hours, or even more frequently.”
Neuropharmacologist Theodore Cicero of the Washington University School of Medicine observed that patients “for whom the drug does not last for 12 hours can suffer both a return of their underlying pain and ’The beginning states of acute withdrawal. That becomes a very powerful motivator for people to take more drugs.’” In a 2001 interview, Dr. David Haddox displayed the stunning lack of empathy typical of Purdue Pharma consultants and executives towards patients who ended up taking OxyContin more frequently than advised, because Oxy failed to relieve their pain for 12 hours. Haddox stated “A lot of these people say, ‘Well, I was taking the medicine like my doctor told me to,’ and then they start taking them more and more and more. I don’t see where that’s my problem.”
Dr. Haddox was a pain specialist on the Purdue payroll, and was one of the most outspoken advocates of prescribing high doses of opioids. Haddox admitted that many people had developed a tolerance to OxyContin, requiring larger doses to achieve the same relief from pain. He also agreed that some patients exhibited classic symptoms of narcotic withdrawal before the end of the 12-hour dosing cycle. However, Haddox denied that these were signs of a developing addiction: in fact, Haddox coined the term “pseudo-addiction” to refer to these symptoms, in an article in Pain. Pseudo-addiction, claimed Haddox, “Seems similar to addiction, but is due to unrelieved pain.” But pseudo-addiction would generally stop when the pain was relieved, “Often through an increase in opioid dose.” In other words, signs of withdrawal and addiction would go away if the patient was given larger doses of opioids! In 2016 a Los Angeles Times investigative report stated that “more than half of long-term OxyContin users were on doses that public health officials considered dangerously high.”

Figure III.2: Dr. David Haddox, a pain specialist who was on the Purdue Pharma payroll. Haddox invented the term ‘pseudo-addiction,’ a highly controversial hypothesis referring to patients who developed symptoms of withdrawal while on OxyContin. Haddox claimed that these were not symptoms of ‘real’ addiction, but simply unrelieved pain; if the dose of opiates was increased, the pain would be relieved and the symptoms would go away.
Of course, Purdue Pharma was enamored with this novel approach to signs of addiction – doctors should just “titrate up,” increasing the dose of opiates, and the problem would go away. In 2015, Green and Chambers performed an investigative search of the medical literature that mentioned “pseudo-addiction.” They found that positive references to this term were generally found in articles or literature supported by the pharmaceutical industry. Furthermore, Green and Andrews found no empirically tested studies to validate or refute this hypothesis. After the horrible experience with OxyContin, it would seem that “pseudo-addiction” should be rejected as a concept, unless it can be supported by rigorous clinical studies.
Purdue Pharma stressed the benefits of moving OxyContin patients to higher doses. In 1996, a Tennessee manager sent a memo to sales reps with the title, “$$$$$$$$$$$$$ It’s Bonus Time in the Neighborhood!” The memo emphasized that “raising dosage strength was the key to a big payday.” However, high doses of powerful opioids naturally lead to addiction and overdosing. The Canadian province of Ontario carried out a study of 32,000 patients on opioid painkillers. They found that “one in 32 patients on high doses fatally overdosed.” Lead researcher David Juurlink said “In other words, they are more likely to die as a result of their medication than almost anything else.”
A related issue was withdrawal symptoms. The position of Purdue Pharma was that, because of the time-release nature of the drug, no “peaks” or “troughs” would be experienced by the user. They further argued that patients could stop taking OxyContin without experiencing withdrawal symptoms. Again, this claim was based on no extensive clinical studies. And many users did experience withdrawal symptoms when they stopped using OxyContin. Not surprisingly, with a powerful opiate, some users began to abuse the drug. Users quickly discovered that if they crushed an OxyContin pill, it would release all of its oxycodone immediately. That wasn’t hard to figure out: the OxyContin package insert warned users that “Taking broken, chewed or crushed OxyContin tablets could lead to the rapid release and absorption of a potentially lethal dose of oxycodone.” So people who got their hands on OxyContin tablets would chew or crush the tablets, and then snort, or dilute and inject, the full dose of oxycodone.
We now know that Purdue Pharma began hearing reports of misuse of OxyContin as early as 1997, just one year after the OxyContin release. In fact, some of their sales reps would report hearing from doctors about people abusing their product. They would suck off the time-release pill coating and snort the pill contents, or cook them, or dissolve the powder and inject it. Purdue’s top lawyer Howard Udell mentioned that the company had “picked up references to abuse of our opioid products on the Internet.” So what was Udell’s response? He “made efforts to limit any written record of concern within Purdue that the company’s wonder drug was being abused.” And the company later insisted that they had never heard any mention of OxyContin abuse before 2000, the year that the head federal prosecutor in Maine sent a letter to doctors all over the state warning them about increasing abuse of OxyContin.
Here, Purdue Pharma was lying. When investigators eventually obtained access to Purdue reports from field representatives, “they found hundreds of references to words like ‘street value,’ ‘snort’ and ‘crush.’” Purdue Pharma had known already in 1997 that patients were abusing OxyContin; more than that, it was easy to bypass the time-release function of the drug, so one could get an immediate hit of a powerful opiate.
Purdue pharmaceuticals even knew that their previous time-release product, MS Contin, had been abused. “In May 1996, an employee sent Richard Sackler and Howard Udell a press report describing the abuse potential associated with users extracting the morphine from MS Contin tablets … In March 1998, Udell sent a memo to Friedman, as well as Mortimer, Raymond, Richard and a handful of other Sacklers, attaching an article from theOttawa Citizen that described how MS Contin had become a street drug in Canada with enough prevalence to have earened a nickname – ’purple peelers’” (the nickname referred to the fact that abusers peeled off the purple coating of MS Contin pills that governed the time-release function; they then crushed the pills, mixed them with water, heated them in a spoon and injected the drug).
Also, a 1998 issue of the Canadian Medical Association Journal contained an article by Dr. Brian Goldman. With respect to MS Contin and other narcotic drugs, Goldman pointed out “The relatively high street price of controlled-release opioid analgesics clearly indicates that these drugs are coveted. This should ring alarm bells.” And Goldman predicted that the newly-marketed OxyContin would be abused in the same way. “Now that controlled-release oxycodone [OxyContin] has been licensed in Canada, we can expect that it and other controlled-release opioid analgesics will also find their way onto the black market.”
What did Purdue do when they realized that their drug was being abused? Perhaps it is easier to list what Purdue Pharma did not do when they realized that OxyContin could rather easily be abused. They did not mention this to the FDA; in fact, their official position (a lie) was that they heard nothing about OxyContin abuse until 2000, when a federal prosecutor in Maine notified thousands of doctors in the state about abuse of that drug. They did not notify pharmacy chains about potential issues with misuse of their drug. They did not notify local authorities in a timely fashion about physicians who were over-prescribing OxyContin. Purdue Pharma went full steam ahead with their legions of sales representatives who provided doctors with the same misinformation about abuse of OxyContin.
The Purdue Pharma management and legal staff took the position that they were offering a great product. They parroted the company line that it was “extremely rare, probably less than 1%” of people who would develop addiction issues when taking the drug under the care of a physician – and they continued to focus their attention on primary-care physicians, the people least likely to have any experience in pain management. Purdue Pharma, and in particular their president Richard Sackler, insisted that any problems resulted from the fact that some of the users of their product had “addictive tendencies.” Thus, they blamed the victim – Sackler recommended that his company “Hammer on the abusers in every way possible.” The addicts themselves were “The culprits … they are reckless criminals.” Purdue Pharma took the position that they were the victims of the “criminals” who became addicted to OxyContin, or who abused that drug. They failed to point out that many of these “criminals” began taking OxyContin under a physician’s care, but sought to overcome withdrawal symptoms by finding ways to increase their dosage, often at the explicit urging of doctors indoctrinated by Purdue Pharma sales reps.
Barry Meier Gets a Tip:
Barry Meier was an investigative reporter. He honed his investigative skills at, of all places, a journal called Chemical Week. Although this was primarily an insider industry journal, Meier pressed forward with exposés about various misdeeds in the chemical industry. Meier’s most famous article for that journal involved revelations that the Dow Chemical plant in Midland, Michigan had leached chemicals containing Agent Orange into the local groundwater. When Meier’s story on Dow Chemical was picked up by the Wall Street Journal, he eventually landed a job as an investigative reporter for the New York Times.
One day in 2001, Meier was given a tip by one of the Times editors, who had heard of a “hot new drug on the street” in the Midwest. It had suddenly become the most popular recreational drug, but paradoxically it was a legally prescribed pharmaceutical called OxyContin.
Meier and his colleague Francis X. Clines began to track down reports of abuse of OxyContin. Purdue Pharma had marketed the drug as “non-abusable,” said a pharmacist. “But that’s not tracking what I’m seeing.” So Clines and Meier submitted a story that appeared on the front page of the Feb. 9, 2001 Times: “Cancer Painkillers Pose New Abuse Threat.” The story stated that “Police detectives in dozens of rural areas in Eastern states are combatting what they say is a growing wave of drug abuse involving a potent painkiller prescribed for terminal cancer patients and other people with severe pain.” Meier and Clines noted that a black market had been established for OxyContin tablets. They identified instances of abuse, overdoses and illegal trafficking of OxyContin in Maine, Kentucky, Ohio, Pennsylvania, Virginia, West Virginia, and Maryland. And over the years, Meier continued to follow this story (see here and here).
What was the response from Purdue Pharma when they got word that reporters were preparing an “OxyContin abuse story”? Purdue head of marketing Michael Friedman suggested a strategy that “Deflects attention away from the company owners”! That is, instead of acknowledging the problem and addressing it, Purdue Pharma tried to prevent people from realizing that the company was run by the Sackler family (the Clines-Meier article had not mentioned the Sacklers). All evidence we have seen from Purdue e-mails and company correspondence at this time reveals a total and shocking lack of empathy for those people who had become addicted to, and even died from, OxyContin. Informed in Feb. 2001 that a single state had registered 59 deaths related to OxyContin, Purdue president Richard Sackler responded “This is not too bad. It could have been far worse.”
Meier continued to follow the OxyContin story. Eventually he was contacted by a Purdue Pharma sales rep, who handed Meier a list of “Toppers,” Purdue’s top ten sales reps in the U.S. The Purdue employee alerted Meier that “every district that was represented on that list was a “hot spot” for OxyContin abuse.” This demonstrated that, of course, Purdue Pharma knew exactly where they were selling the most pills. And they had to know that OxyContin was selling most rapidly in areas where the drug was causing addiction and being abused.
A Black Market for OxyContin:
In these areas, a black market in OxyContin pills had developed and was expanding. First, Purdue Pharma sales reps would descend on a community, touting OxyContin as the “cure” for chronic pain, including back pain, knee pain, or fibromyalgia. Primary-care physicians would prescribe OxyContin to their patients. For several patients, Oxy would not relieve their pain for 12 hours, and a considerable number of these patients would develop a tolerance to OxyContin. For a while this could be allayed by taking the drug more frequently, perhaps three or four times per day; and/or they could be prescribed OxyContin tablets with higher doses of oxycodone. However, these users would eventually reach the point where their doctors would no longer keep upping their doses. At that point, the users had become addicts; the opiates had overwhelmed their brain receptors, and their lives revolved around obtaining “hits” of opioids. The users would travel to several doctors, in efforts to obtain multiple prescriptions of OxyContin. They would crush the tablets and either snort or inject the drug, which provided an immediate rush.
However, OxyContin was not cheap, and users needed a steady income to feed their habit. So a black market in OxyContin developed. Soon OxyContin tablets were selling for $1 a mg. For the addicts, this was a losing game, as every dollar they made selling Oxy tablets went to feed their habit. However, in areas with a black market, seniors discovered that they could get a prescription for OxyContin simply by claiming they were in severe pain. Seniors on Medicaid merely had to pay a monthly $3 fee that covered the cost of all prescribed meds for that month. Seniors who were prescribed 80-mg doses of OxyContin would be able to sell the pills for $80 each (the areas of the country with the largest black markets also had the highest percentage of 80-mg OxyContin being prescribed).
On the illegal-drug market, drug dealers began to replace other illegal drugs with OxyContin. Dealers who had previously dealt in meth or cocaine now frequently shifted to OxyContin, which was more profitable for them. It is not clear to what extent the sales reps understood the nature of the black-market problem, although some of the “Toppers” sales reps heard from doctors and pharmacists that OxyContin was being abused, and some reps conveyed this to their Purdue bosses. The top corporate officials, marketing heads and lawyers at Purdue Pharma knew that there was a problem, but they continued pushing sales of OxyContin, paying out unheard-of bonuses to their top sales reps.
In his 2015 book Dreamland, Sam Quinones attempted to discover “Doctor Zero,” the first doctor to start a “pill mill.” Quinones claims that Dr. David Procter quite likely opened the first pill mill in Portsmouth, Ohio, a small town on the Ohio River adjoining the borders of Ohio and West Virginia. In 1997, Procter had opened a pain-management clinic in Portsmouth. He specialized in prescribing OxyContin to his clients. His business exploded with clients. Neighbors of Procter’s clinic reported seeing vast numbers of patients, with lines overflowing from his office down the street. Some patients would arrive in their cars and wait for hours. Some of the license plates were from out-of-state locations as far afield as Missouri or Arkansas.

Figure III.3: Dr. David Procter. Sam Quinones, author of Dreamland, believes that Procter may have run the first “pill mill” that recklessly over-prescribed drugs, particularly OxyContin, to drug abusers.
As far as one knows, Procter began as the proprietor of a legitimate pain-management clinic. Initially, he counseled patients about physical therapy and other pain-management strategies, and he referred patients to other local doctors. But at some point, it appears that Procter himself became habituated to OxyContin. From then on, his practice became almost exclusively focused on hard drugs. In addition to drugs containing opiates Vicodin (5 mg hydrocodone + 325 mg acetaminophen) or OxyContin, Procter also recommended benzodiazepines (psychoactive drugs such as Valium and Xanax), often prescribed together with OxyContin. Both categories of drugs were depressants, and taking them together was quite dangerous; but it was reported that “the benzos seemed to enhance the euphoria of the opiates.” Procter’s business now became cash-only. According to Quinones, his clinic became “a virtual ATM for dope.” He saw hundreds of patients a day, prescribing opiates and other meds to all of them. Several women claimed that he traded prescriptions for sex with them.
Procter had practices in both Portsmouth, Ohio and across the Ohio River in Kentucky. As his standards slipped, the number of complaints against him increased. He became a target of medical licensing boards in both Kentucky and Ohio. Medical inspectors referred to his practice using terms such as “gross incompetence,” “negligence,” and “malpractice.” They commented on his work, “There is significant lack of evidence of evaluation and alternative treatment to this patient except for the use of controlled substances.” At the end of 1998, Procter closed his practice in Kentucky but maintained his pain clinic in Portsmouth. Over the next three years, Procter hired fifteen different doctors to assist in his clinic. “They arrived with histories of drug use, previously suspended licenses, and mental problems, ready to prescribe while working for Procter for $2,500 a week.” Many of these doctors, having participated in a pill mill, went on to open their own pill mills; in fact, one law official referred to Procter as the “Ray Kroc” of the pill mill business (after Kroc, who franchised McDonald’s restaurants across the country).
Sam Quinones mentions one of Procter’s doctors, Frederick Cohn. Cohn opened a pain clinic in Paintsville, Kentucky. Cohn “saw as many as 146 patients a day, three minutes each, while lines formed outside. Cohn had preprinted prescriptions for various narcotics, including OxyContin, Lortab (10 mg hydrocodone + 325 mg acetaminophen), Soma and Xanax. He prescribed the same pills in the same amounts over and over throughout the day, no matter what the patient’s complaint – 2.7 million pills in one year.”
Pain Killer:
Barry Meier wrote a book about OxyContin called Pain Killer: A “Wonder” Drug’s Trail of Addiction and Death.” It was published in fall 2003, and it represented a major work of investigative journalism. The book arose from Meier’s New York Times stories on the impact of OxyContin. And it properly laid the blame for a mounting trail of addiction and death to the reluctance of Purdue Pharma to take any responsibility. Meier claimed that Purdue “Seemed unable or unwilling to take dramatic action until long after circumstances or adverse publicity had forced their hand.” In fact, Purdue Pharma had continued its policy of admitting nothing and contesting everything by sending a delegation of officials to the New York Times to complain that Meier’s reporting for that paper was “sensationalized and skewed.” At that time, Meier’s editor stood firm and defended his reporting.
However, after Pain Killer was published, their head lawyer Howard Udell and other Purdue Pharma officials returned to the Times with a new complaint. They now claimed that Meier, having written a book about Purdue Pharma, should be prohibited from writing any more Times articles about their company. They claimed that his book constituted a conflict of interest for Meier: Meier would be unable to write fair-minded articles about Purdue Pharma because of the prejudice he had shown in his book. To the Times’ everlasting discredit, they accepted this astounding piece of chutzpah by Purdue Pharma, and forbade Meier to write any more articles about Purdue or OxyContin. If you follow Purdue Pharma’s reasoning, then every reporter who wrote a book would afterwards be forbidden to file stories on that topic.
Purdue Gets Hauled Into Court:
After Barry Meier’s book was published, Purdue Pharma was bombarded with lawsuits by people who had become addicted to OxyContin. However, the company’s policy, under their combative lead lawyer Howard Udell, was to contest every lawsuit and admit nothing. The company’s defense was that Purdue Pharma were the victims in this process; people only became addicted if they had “addictive tendencies,” and Purdue’s fine product was being mis-used by these abusers. The company took advantage of the fact that the people suing them had been suffering from long-term chronic pain before starting OxyContin. If Purdue Pharma could show that the plaintiffs had any earlier issues with over-use of prescription drugs or alcohol, the company would use this as proof of “addictive tendencies.” Also, once these people became addicted, they would resort to crime in order to feed their habit; again, Purdue Pharma could vilify the plaintiffs as criminals.
By 2002, Purdue was spending $3 million per month on lawyers fighting litigation. In 2003, Purdue’s head lawyer Howard Udell issued a press release with the heading “65-0.” At that point in time, that was Purdue’s record in defending lawsuits. One would think that 65 lawsuits for causing addiction would have made an ethical company think twice about its business model; but Purdue Pharma had no such qualms. The company also went out and contracted with several consultants with records in government service. These “hired guns” included Howard Shapiro, who was the former general counsel to the FBI and later a partner at Wilmer Cutler Pickering Hale and Dorr. They also included Eric Holder, who had been a deputy attorney general under Clinton; and they hired former U.S. attorney Mary Jo White; and they also hired Rudy Giuliani, who was apparently coordinating the actions of the other defense attorneys.

Figure III.4: Mary Jo White. She had been a former U.S. Attorney; after that, she would be appointed by President Obama to head the SEC. She has represented Purdue Pharma or various members of the Sackler family since 2005.

Figure III.5: Howard Shapiro. He was the former general counsel to the FBI. He was hired by Purdue Pharma to defend them in a 2007 lawsuit. Over the years, Shapiro’s law firm was paid over $50 million for his work for Purdue.
John Brownlee was the U.S. Attorney for the Western District of Virginia. That Appalachian area had long been plagued by drug and alcohol abuse. But Brownlee was now hearing about a new plague of addiction that seemed to be centered around abuse of OxyContin. So Brownlee began to mount an investigation of the makers of OxyContin, Purdue Pharma. As part of his preliminary investigations, in Dec. 2002 Brownlee requested that Purdue send him copies of their documents relating to the manufacturing, marketing and distribution of OxyContin. Brownlee’s team of investigators knew that companies often tried to overwhelm litigants by burying them in information. Purdue Pharma was no exception: eventually they sent millions of pages of documents to Brownlee’s team.
It took the team quite a while to sift through all that material, but when they were done the legal team had myriad copies of “e-mails and memos and meeting minutes and marketing plans from the company.” In addition, they had notes filed by Purdue field reps from all over the country. The investigators concluded that “nearly every major element of the story that Purdue had been telling about its own conduct was false.” Not only did Purdue lie about their lack of knowledge of any abuse of OxyContin, the company even knew about significant abuse of their earlier drug MS Contin. Purdue’s own internal studies of OxyContin showed that almost 30% of patients in one study reported experiencing withdrawal symptoms when they stopped using the drug (at very low doses). Purdue had also carried out experiments to see how much oxycodone could be extracted if the pills were dissolved (answer: most of it).
Despite this, Purdue Pharma executives had testified under oath that until 2000, they had no idea that OxyContin was being abused. And despite their own experiments dissolving OxyContin in liquid, they trained their sales reps to tell doctors their pill could not be injected. Brownlee and his team had videotapes of the training of sales reps, in which claims were made that the company knew for a fact were untrue. And Purdue was sending sales reps to visit doctors, even when they knew those doctors were under investigation or their licenses had been suspended.
Slowly but surely, Brownlee and his team assembled their information about Purdue Pharma and its executives. In Sept. 2006, one of the prosecutors Rick Mountcastle assembled a confidential prosecution memo laying out their case. Their intention was to bring multiple felony charges against three of the top executives at Purdue: president Michael Friedman (Richard Sackler had stepped down as president in 2003, although this was simply for show: Sackler continued to run the company behind the scenes); Purdue’s top lawyer Howard Udell; and Dr. Paul Goldenheim. They were to be charged with misbranding, wire fraud, mail fraud, and money laundering. Purdue Pharma would also be charged with multiple felonies. And the attorneys decided to ask for a $1.6 billion fine against the company.
Brownlee and his staff were confident that their allegations were supported by a mountain of evidence from the company itself. Their prosecution memo detailed “the story of an intricate, years-long, extraordinarily profitable criminal conspiracy.” The Sackler family was not mentioned in the allegations; however, Brownlee and his team felt that under the weight of numerous felony allegations, at least one of the three executives was likely to agree to a plea agreement by naming one or more of the Sacklers. In this way, Brownlee et al. might hope to “work their way up the corporate ladder,” eventually reaching the Sacklers.
But in October 2006, John Brownlee and his team were summoned to a meeting at the Justice Department. That meeting was chaired by assistant attorney general Alice Fisher, and included political appointees in the George W. Bush administration. In addition, it included the three Purdue executives that Brownlee had intended to charge with felonies, and also Purdue legal consultants Howard Shapiro and Mary Jo White. Both Purdue and the Brownlee team made arguments about the case. Shortly afterwards, Brownlee was informed that the Justice Department would not support felony charges against the Purdue executives. Instead, Purdue would plead guilty to one count of felony misbranding (that is, that pharmaceutical products had been mislabeled); the three executives would each plead guilty to one misdemeanor count of misbranding.
Eventually, additional charges and penalties would be assessed. But this occurred only after Mary Jo White phoned Alice Fisher’s boss Paul McNulty to ask that even the misdemeanor charges against the Purdue executives be dropped. Eventually Brownlee gave an ultimatum: if Purdue and the executives did not sign off on guilty pleas, he would proceed with criminal charges against the executives. At this point, Purdue agreed to plead guilty to one felony charge of misbranding and to pay a fine of $600 million. Udell, Friedman and Goldenheim would plead guilty to misdemeanor misbranding. They were barred from doing business with any taxpayer-funded health-care program for 20 years, and they were assessed fines totaling $34 million (those fines were paid by Purdue).
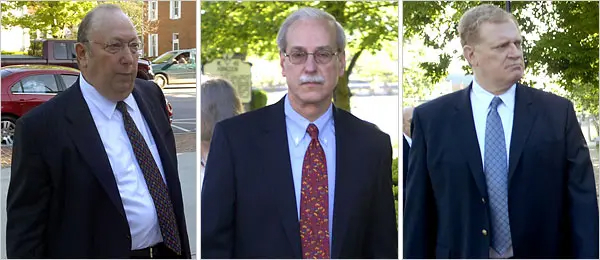
Figure III.6: Purdue Pharma executives (from L) Howard Udell, Paul Goldenheim and Michael Friedman. They each pleaded guilty to one misdemeanor count of misbranding, and were barred from doing business with any taxpayer-funded health-care program for 20 years.
Brownlee might have consoled himself that, although he had failed to convict any of the executives of the multiple felonies that he had contemplated, he had shut down the marketing of OxyContin. If Purdue Pharma pleaded guilty to a felony, the company would be barred from doing business with taxpayer-funded health agencies, such as Medicare and Medicaid. That would have put OxyContin out of business, and shut down this drug that had kick-started a terrible legacy of addiction and death. But wait! – Purdue’s attorneys pulled one last rabbit out of their hat. The company that pleaded guilty to a felony was not Purdue Pharma: it was Purdue Pharma’s parent company, Purdue Frederick! This was the company that had manufactured laxatives and earwax. After the plea, Purdue Frederick went out of business.
Business As Usual:
But Purdue Pharma and OxyContin? They continued much as before. Not long after the guilty plea, Purdue Pharma hired 100 additional sales representatives. And the company would provide Howard Udell with a $5 million payout and Michael Friedman with $3 million (a practice sadly reminiscent of Mafia dons paying off lieutenants for ‘taking the fall’). One would have thought that after admitting to felony misbranding of its product, executives at Purdue Pharma would modify their tactics and stop the reckless sales of such a dangerous opiate. But at Purdue’s corporate headquarters, it was mainly business as usual. Although Purdue Pharma took some steps to curb the abuse of OxyContin, they continued the same sales tactics and the same lies told by their sales reps (addiction is extremely rare, OxyContin can’t be abused, there are no ‘peaks’ or ‘troughs’ because of the time-release mechanism, withdrawal symptoms are simply ‘pseudo-addiction’ that can be cured by higher doses of the opiate). Purdue Pharma continued manufacturing OxyContin as fast as possible, and targeting doctors who sold enormous quantities of their pills, in large part regardless of the ethical implications.
The Los Angeles Times covered a particularly flagrant case in LA, where a criminal ring set up an elderly doctor named Eleanor Santiago in a pain clinic. The crime ring would round up homeless people and pay them $25 to attend the clinic for an ‘examination’ and an 80 mg OxyContin prescription. The patients would then be driven to a pharmacy; the criminals would take the Oxys from the indigents and distribute them to black markets all over the Western U.S. Dr. Santiago prescribed over 73,000 OxyContin 80-mg pills in less than a year. The Purdue district manager who visited Santiago’s ‘clinic’ told Jack Crowley, the compliance officer at Purdue Pharma, that the clinic looked like a drug ring; she asked “Shouldn’t the DEA be notified?” Purdue never mentioned this to law authorities until 2010, by which time the clinic had been shut down and Dr. Santiago indicted. During the two years of Santiago’s clinic, Purdue Pharma supplied her with over a million OxyContin pills. “Crowley would later acknowledge that in the five years he spent [as Purdue’s compliance officer], the company did not suspend the flow of pills to a single” suspicious doctor or pharmacy.

Figure III.7: Dr. Eleanor Santiago. A crime ring installed her as head of a phony pain clinic called Lake Medical. During the two years of operation of Lake Medical, Purdue Pharma supplied Dr. Santiago with over a million OxyContin pills.
It is hard to characterize the actions of Purdue Pharma, under the leadership of the Sackler family, as anything other than depraved indifference to human life. A nationwide opioid crisis, of which OxyContin was the driving force, was sweeping the country. They had been convicted of felony misbranding of Oxy, yet they continued to press ahead with the same sales pitches that they knew were false and lacked any corroborating evidence. In 2007, following the guilty plea by Purdue Frederick, Purdue Pharma engaged the McKinsey consulting firm to advise how they might continue to grow sales of OxyContin.
McKinsey made aggressive suggestions to Purdue on how they could “turbocharge” sales of OxyContin. They recommended “Sales ‘drivers’ based on the idea that opioids reduce stress and make patients more optimistic and less isolated.” They also consulted on ways to “Counter the emotional messages from mothers with teenagers that overdosed” on Oxy. In 2013, the federal government reached a settlement with the pharmacy chain Walgreens to crack down on illegal opioid prescriptions. McKinsey recommended that Purdue “Lobby Walgreens’ leaders to loosen up.” In other words, Purdue should attempt to prevent Walgreens from limiting illegal sales of opioids!
This was not the only outrageous suggestion from McKinsey consultants. They also recommended giving pharmacies a rebate for every OxyContin overdose attributed to pills that the pharmacy had sold. McKinsey even “estimated that 2,484 CVS customers would overdose or develop an opioid use disorder in 2019 from taking OxyContin.” So McKinsey knew precisely the dangers of the drug whose sales they were attempting to “turbocharge”!
In 2010, Purdue Pharma released a new pill, OxyContin OP. They had re-formulated the cover of OxyContin, so that it was much harder to crush the pill and release all of the oxycodone at once. Purdue Pharma touted this as a great step forward. Indeed, it was a lot harder to defeat the new coating, and if you succeeded, the resulting “hit” of oxycodone was much smaller. One immediate question was why the FDA had not required Purdue Pharma to do this when they first approved it. Had they done this, much of the resulting American opioid crisis might have been averted. But Purdue Pharma also knew that the most prevalent way that OxyContin was abused was simply by taking more pills than the recommended dose of two per day. The new formulation did nothing to lessen this method of abuse.
Unfortunately, by 2010 the U.S. was in a full-blown opioid crisis. And this was now exacerbated by a new entry into the drug abuse market. Actually, this ‘new’ drug was one of the original illegal opioids – heroin. But this was a new twist on the powder heroin that had previously dominated the market. Sam Quinones details how a deadly new scourge came to be marketed in the U.S. The small Mexican state of Nayarit grows copious amounts of opium poppies. And they learned to cook this into “black tar” heroin. Families in Nayarit harvested and processed the heroin, and then smuggled it into the U.S.

Figure III.8: The state of Nayarit, on Mexico’s western coast.
The product would then be cut up in chunks that were encased in small balloons. Then, dealers would locate promising areas in the U.S. The most likely places were towns with significant numbers of people on disability, or areas with pain clinics and/or methadone clinics. The Nayarit cartel would then recruit youths from Mexico to sell the drug. The youth were highly unusual in that none of them used the heroin. Rather than store large quantities of heroin in houses that could be raided, each courier worked out of his car, and carried only small amounts of heroin, in balloons that could be swallowed if necessary. Orders were taken by cell phone, and the drivers delivered door-to-door, just like a pizza delivery. Since none of those involved in the delivery chain used heroin, the black tar heroin arrived uncut, many times stronger than the traditional powder heroin that was typically cut by everyone who handled the drug.

Figure III.9: Samples of black tar heroin, the ‘baggies’ in which they were placed, and other drug paraphernalia.
As a general rule, the Nayarit cartel sold only to whites, as they feared the violence they associated with the black drug culture. In addition, their drivers never carried weapons, since the penalty for being caught with small amounts of drugs was deportation, whereas drugs plus weapons would bring a ten-year sentence. One frightening aspect of the Nayarit cartel was that they were able to sell their black tar heroin at a significantly lower price than the black-market rate for OxyContin. Thus, many people who were addicted to OxyContin subsequently switched to heroin. Another selling point of the black tar heroin was that it could be smoked. Some addicts had an aversion to injecting drugs – although most addicts would find that their reluctance to shoot up was overwhelmed by their need to score a hit.
Around 2010 the American prescription-drug abuse epidemic morphed into a heroin epidemic. The executives at Purdue Pharma would point to this as proof that their assertions were correct – the only people who developed a drug problem were those with pre-existing “addiction tendencies.” However, the American Society of Addiction Medicine found in a study that about 80% of people who began using heroin around 2010 had begun by abusing prescription painkillers. In 2019, a paper titled “How the Reformulation of OxyContin Ignited the Heroin Epidemic” detailed the “rapid rise in the heroin death rate” immediately following the release of the reformulated OxyContin OP.
After 2010, Purdue Pharma was continuing to enjoy phenomenal sales of OxyContin: over two decades the drug brought in some $35 billion in sales. However, at the same time Purdue was fending off legal suits over the company’s role in causing a deadly surge of opiate addiction in the U.S. Anticipating eventual losses in court, and a decline in demand for OxyContin, they turned to their international arm, Mundipharma. The Sacklers saw great potential for repeating abroad what it had done in the U.S. They began to spread their “new pain management” message abroad.
Mundipharma moved into Mexico in 2014, where their representatives announced that 28 million Mexicans were living with chronic pain. This “pain epidemic” apparently existed in many other countries as well – Mundipharma claimed that 80 million Brazilians were also suffering from chronic pain, and 22 million Colombians (47% of the country’s population) were also living in untreated pain. Mundipharma also sponsored visits by doctors on its payroll, who were flown all around the world to repeat the baseless claims they had made in the U.S. Mundipharma re-cycled the demonstrably false claims they had made in the U.S. – that their time-release formula made it “almost impossible for those with chronic or severe pain to become addicted.”
In 2007, the state of Kentucky charged Purdue Pharma with deceptive advertising of their product. The trial finally was scheduled to begin in the fall of 2015. However, at the last minute Purdue Pharma agreed to pay $24 million to Pike County, Kentucky, a region that had been completely devastated by a raging opioid addiction problem. Although this was much larger than Purdue Pharma’s opening offer of $500,000, it was a drop in the bucket compared to the magnitude of the addiction problem, jump-started by Purdue’s release of OxyContin. Polls showed that 29% of the inhabitants of Pike County knew someone who had died from using OxyContin, and 70% of the respondents to a survey agreed that Purdue Pharma had a “devastating” effect on their county.
But the resolution of the case had a “golden lining” for Purdue. Under the terms of the agreement, the company admitted to no wrongdoing. Even more important, as part of the agreement, all of the millions of pages of evidence amassed by the state of Kentucky were to be sealed forever from public view. So, Purdue Pharma paid $24 million, but in the process were allowed to go on marketing their product. And their expectation was that no one else would ever see the evidence against them.
The Hammer Falls on Purdue Pharma:

Figure III.10: A protest against the Sackler family and their marketing of OxyContin.
However, the exponential growth of the opioid crisis eventually led people to connect the dots and to realize the extent to which Purdue Pharma had driven the opioid crisis. The L.A. Times broke a series of major stories about the company and its practices. In 2013 it disclosed Purdue’s “Region Zero list.” This was a database containing some eighteen hundred doctors who were suspected of operating “pill mills.” The Times reported that the company compiled the database of doctors “recklessly prescribing its pills to addicts and drug dealers, but has done little to alert law enforcement or medical authorities.”
Then in 2016 the L.A. Times published another big story. In this case they reported that Purdue Pharma had realized, even before releasing OxyContin, that it would not relieve pain for twelve hours. Apparently, patients in clinical trials had complained that their pain returned before twelve hours. But Purdue nevertheless maintained that OxyContin would relieve pain for twelve hours. The fact that patients resorted to taking the pills more frequently, in order to relieve their pain, was a first step in what could easily develop into an addiction.
By this time, “more than 165,000 Americans had lost their lives to prescription drug abuse since 1999. Overdoses now surpassed car accidents to become the leading cause of preventable death in America.” Surveys showed that “nearly half of all Americans knew someone who had become addicted to prescription opioids.” At this point, the reputation of Purdue Pharma had become toxic. But this caused a schism within the company. The “old guard,” primarily long-time staff personally associated with the Sackler family, advocated continuing with the company’s policy of conceding nothing and challenging everything. Newer staff and executives suggested several alternatives, including establishing a foundation to address the opioid crisis and diverting some of their philanthropic efforts in the arts toward addiction centers. Purdue Pharma rejected all of these suggestions.
When challenged about the dangers and widespread abuse of OxyContin, Purdue Pharma would invariably point to the fact that the FDA had approved their product. And for a while, FDA officials had supported Purdue Pharma’s claims. However, as the opioid crisis raged and the claims made by Purdue Pharma began to unravel, support from the FDA began to wane. David Kessler, who had been head of the FDA when OxyContin was approved, described the approval of heavy-duty opiates as one of the “great mistakes of modern medicine.” But still, the FDA did relatively little to combat the widespread prescribing and abuse of opioids.
In 2011, the Centers for Disease Control described the crisis of addiction and death that was sweeping the country as an epidemic. CDC Director Tom Frieden said that even the reformulation of OxyContin had been dangerous because it reinforced the perception that these opioids were safe. “People thought it was less addictive, but that was a big distraction,” said Frieden. “The company knew damn well what it was peddling, and I think that’s the right word – peddling.” The CDC began a process to create guidelines that physicians could use, in an attempt to stop the rampant over-prescribing of opiates.
Even Dr. Richard Portenoy, the former “king of pain,” acknowledged second thoughts about his former assertions. “Did I teach about pain management, specifically about opioid therapy, in a way that reflects misinformation? I guess I did.” Portenoy told a Wall Street Journal reporter that his earlier lectures about opioids and addiction “weren’t true.” Further, he admitted that “Data about the effectiveness of opioids does not exist.”
In 2018 Mike Moore, who had previously been the attorney general of Mississippi, began to coordinate a multi-state coalition of lawsuits against Purdue Pharma and the Sackler family. Moore had been a veteran of such lawsuits against Big Tobacco, and also against BP in the Deepwater Horizon oil spill. To be sure, Moore wanted a cut of the ludicrously large payouts to lawyers in such class-action lawsuits. However, the attorneys general of the many states who joined in the lawsuit were also seeking compensation for their citizens, for their cities and counties overrun with addiction, and for treatment centers.
The intention was to go after all of the many players who had contributed to this plague of addiction. There were pharmaceutical firms, including not only Purdue Pharma but also companies such as Johnson & Johnson and Endo, who also marketed opiates. Manufacturing firms such as McKesson, Cardinal and Amerisource Bergen supplied the drugs to pharmacies, and the large pharmacy chains such as Walgreens, Walmart and CVS filled millions of prescriptions, including issuing drugs to pill mills in the towns where they were located. Purdue Pharma had to realize that they were in big trouble when one of the witnesses against them and their co-defendants was none other than Russell Portenoy. In return for being dismissed as a defendant, Portenoy now stated that he had previously denied the risks of opioids even though he knew of “serious opioid-related adverse outcomes” by the late 1990s. And Portenoy singled out Purdue Pharma, saying that “No other company had previously promoted an opioid drug as aggressively, or encouraged the use of an opioid by non-specialists.”
In Jan. 2019 Maura Healey, the attorney general of Massachusetts, filed a brief against Purdue Pharma; and this time, the complaint named eight members of the Sackler family as defendants. But before Healey had filed the suit naming the Sacklers, she was visited by none other than Mary Jo White, who brought along a phalanx of lawyers in an attempt to convince Healey not to name the Sacklers as defendants.
Healey had an ace in the hole. The multi-district legislation against Purdue Pharma had won the right to view millions of pages of previously sealed court records relating to Purdue and other Big Pharma companies. The judge ruled that those records could not be released to the public, but they were to be made available to the attorneys who had filed suit against the company. As Purdue Pharma knew full well, once the attorneys got their hands on these documents, it would no longer be possible for the Sacklers to argue that they merely served on the board of the company, but otherwise were not involved in the operations of the firm. In fact, the documents made it clear that the Sacklers, and particularly Richard Sackler, made most of the decisions at Purdue. For example, the McKinsey Company who were consulting with Purdue stated that the Sackler family “were involved in all levels of decision-making on a weekly basis.”
Maura Healey then attempted to make public her complaint. Purdue fought vigorously against this, as the complaint (which ran to 274 pages) was filled with material contained in the documents that weren’t supposed to be released to the public. But Healey received permission from a judge in Massachusetts to release her complaint. And Healey’s documents completely destroyed the Sacklers’ mission to remain out of the public eye. The documents showed that even after the CDC’s guidelines on limiting opioid prescriptions to reduce the probability of addiction, the Sacklers pressed their company “to persuade doctors to put patients on higher doses of opioids for longer periods of time.”
Some of the more damning charges in Healey’s complaint showed that, well after the company had pleaded guilty to felony misbranding of their product, they continued to call on doctors who were clearly over-prescribing. One example was Dr. Fathalla Mashali, who ran pain clinics in Massachusetts and Rhode Island. Purdue Pharma knew that Mashali was under investigation by authorities in Rhode Island; nevertheless, they instructed their sales reps to continue visiting his Massachusetts clinics. A Purdue sales rep who visited one of Mashali’s clinics in 2013 reported that the clinic office was so crowded that some patients brought “their own ‘beach type’ folding chairs to sit on because at any given time, he might have 35 or more patients waiting.” Mashali “eventually lost his medical license, pleaded guilty to twenty-seven counts of health-care fraud, and was sentenced to seven years in prison.”
During the period 2008 to 2012, Purdue’s top prescriber in Massachusetts was Dr. Walter Jacobs. He had a one-man practice, but Healey noted that “He often worked only three days a week. Nevertheless, in five years he prescribed more than 347,000 pills of Purdue opioids.” Of those, 200,000 were Oxy 80-mg pills. “Purdue offered Jacobs a $50,000 contract to give speeches.” Healey’s complaint described the company’s practices, “Purdue took advantage of addiction to make money. For patients, it was a massacre.” Of those who died from addiction related to taking OxyContin, “The oldest died at age 87. The youngest started taking Purdue’s opioids at 16 and died when he was 18 years old.”
New York attorney general Letitia James had also filed suit against Purdue Pharma, and once again named Sacklers who were on the company’s board as defendants. James pointed out that immediately following Purdue Frederick’s guilty plea for felony misbranding, the Sacklers began a systematic campaign to siphon funds from the company to themselves; they then transferred much of that money to offshore accounts. James pointed out that from 1997 to 2007, Purdue Pharma had distributed $126 million to the Sacklers. But since 2008, it had distributed billions to the Sackler family.
The Sackler family had always maintained a strange dichotomy when it came to publicity. In their corporate work, they went to immense lengths to avoid publicity. They almost never gave interviews, and they tried to hide their ownership of and involvement in their companies. On the other hand, when it came to their philanthropic gifts, they went to enormous lengths to trumpet their names. Patrick Radden Keefe points out that in the United Kingdom alone this resulted in the “Sackler Building at the Royal College of Art, the Sackler Education Centre at the Victoria and Albert Museum, the Sackler Room at the National Gallery, Sackler Hall at the Museum of London, the Sackler Pavilion at the National Theatre, the Sackler Studios at the Globe Theatre.” And at the Tate Modern museum, “a silver plaque informs visitors that they happen to be riding on the Sackler Escalator.”
As the opioid crisis escalated, the central role of Purdue Pharma in the opioid epidemic became known to the public. Articles about Purdue Pharma that highlighted the role of the Sackler Family appeared in Esquire, the New Yorker and Vanity Fair. Suddenly the veil hiding the secretive Sackler family was lifted. At this point the Sacklers suddenly became a widely despised family.

Figure III.11: Nan Goldin’s group PAIN protesting at the Guggenheim Museum, which had accepted philanthropic gifts from the Sackler family. The protesters dropped slips of paper to represent the OxyContin prescriptions that generated the profits used by the Sackler family in their gifts.
AIDS activist and photographer Nan Goldin had previously entered rehab for a heroin addiction. She was then prescribed OxyContin to deal with severe pain from tendinitis. She soon developed a dependence on Oxy that required another stint in rehab in 2017. She got a first-hand look at the opioid epidemic that was fueled by prescription opiates, so Goldin created an activist group called PAIN (Prescription Addiction Intervention Now). That group staged protests at several institutions that had accepted philanthropic donations from the Sackler family. They organized a protest at the Sackler Wing of New York’s Metropolitan Museum of Art, where they threw pill bottles with the label “OxyContin: Prescribed to you by the Sacklers.” Another protest occurred at the Guggenheim Museum, where protesters stood on the curving balcony, unfurled a large banner, and dropped slips of paper to represent Purdue Pharma “prescriptions.” Other protests occurred at the Louvre, at London’s Serpentine Sackler Gallery, at Harvard’s Sackler Museum, and at the Purdue Pharma headquarters.

Figure III.12: A protest at Purdue Pharma headquarters. Protesters brought in a gigantic bent spoon, as is used by heroin addicts, to highlight Purdue’s role in creating America’s opioid epidemic.
The Sackler family’s long efforts to shield themselves from public scrutiny had now completely backfired. As their role in marketing OxyContin and the wealth they had derived from the opioid epidemic became widely recognized, the Sacklers became the subject of several news investigations. Inevitably, this turned the Sacklers into targets for late-night hosts. Stephen Colbert did a riff on the Sackler family, claiming that their revised version of the Hippocratic Oath went “First, do no harm. Unless harming is incredibly profitable.” John Oliver devoted a segment of his program Last Week Tonight to the opioid crisis. After a succinct and accurate review of the marketing of OxyContin, Oliver focused on the Sackler family’s penchant for privacy. He showed a transcript of a deposition by Richard Sackler and complained that the Sacklers had managed to prevent the video of the deposition from being released. So Oliver recruited a number of famous actors, including Michael Keaton and Bryan Cranston, to perform dramatic readings of Richard Sackler’s testimony. Here is the video of Oliver’s show.
By fall 2019, nearly every state was suing Purdue Pharma, and about half the states included the Sackler family as defendants. So the Sackler family made an offer, to settle all of the suits together. The Sackler family would relinquish control of Purdue, and the company would be turned into a public trust. The family would donate a large sum of money that would be used to alleviate the opioid crisis. The Sackler family would be granted immunity from “all potential federal liability” related to OxyContin; and the family would admit to no wrongdoing. The offer was described in the press as “Purdue Pharma Offers $10-12 Billion to Settle Opioid Claims.”
However, anyone offered a deal by the Sacklers would be advised to review the details with great care. Under scrutiny, the Sackler offer appeared much less generous than at first sight. For one thing, the offer included $4 billion worth of new drugs to treat addiction; the value of those drugs would be counted as an in-kind gift. Furthermore, the offer included $3 to $4 billion in drug sales from the re-constituted Purdue Pharma. Finally, what was counted as $4.5 billion from the Sackler family was contingent upon the sale of Mundipharma for at least $3 billion, in which case the family would cough up an additional $1.5 billion (note that between 2008 and 2016, the Sackler family had paid itself $4.3 billion in donations from Purdue Pharma).
In September 2019, Purdue Pharma declared bankruptcy. Corporations have the ability to pick the bankruptcy judge and they picked Robert Drain. Apparently, Purdue Pharma was counting on Drain to make a series of rulings that greatly favored the Sackler family in these proceedings. For example, although Purdue Pharma was declaring bankruptcy, the Sacklers were not. But Drain seemed to reject the proposal to claw back some of the money the Sacklers had withdrawn from Purdue Pharma. The Sacklers were counting on the fact that they had siphoned so much money from the company that Purdue Pharma was currently worth only about $1 billion.
On November 24, 2020, just weeks after the presidential election, Purdue Pharma pleaded guilty in federal court to one felony count of conspiracy to violate the Food, Drug and Cosmetic Act, and two felony counts of conspiracy to violate the Federal Anti-Kickback Statute. The feds announced that Purdue Pharma had been levied a criminal fine of $3.544 billion and an additional $2 billion in criminal forfeiture. The company agreed to pay $225 million of that fine immediately. In addition, the company also agreed to an “unsecured bankruptcy claim” of $2.8 billion “to resolve its civil liability under the False Claims Act.” And the Sackler family agreed to pay $225 million in damages to resolve its civil False Claims Act liability. Going forward, Purdue Pharma would emerge as a “public benefit company (PBC)” and the remaining $1.775 billion of the $2 billion forfeiture would be credited to funds generated by the PBC.
On July 8 2021, 15 states reached an agreement with Purdue Pharma. Under this agreement, Purdue PHarma will contribute $500 million, as soon as the company emerges from bankruptcy. Also, the Sackler family will contribute a total of $4.5 billion. Of this amount, a national opioid abatement fund will oversee Sackler charitable arts trusts worth at least $175 million. All of those funds will go towards addressing the opioid crisis.
The Sackler family has insisted that, in return for their contributions, they be released from any further lawsuits. The states that have signed onto this agreement were: Colorado, Hawaii, Idaho, Illinois, Iowa, Maine, Massachusetts, Minnesota, Nevada, New Jersey, New York, North Carolina, Pennsylvania, Virginia and Wisconsin. The following states continue to oppose the plan: California, Connecticut, Delaware, Maryland, New Hampshire, Oregon, Vermont and Washington, also the District of Columbia. The states opposing the plan cite the fact that under this agreement, the Sackler family have “misused this bankruptcy to protect their vast wealth and evade consequences for their callous misconduct,” in the words of Connecticut attorney general William Tong. The opposing states will attempt to extract more money from the Sackler family.
On the other hand, New York attorney general Letitia James stated, “While this deal is not perfect, we are delivering $4.5 billion into communities ravaged by opioids on an accelerated timetable and it gets one of the nation’s most harmful drug dealers out of the opioid business once and for all.”
Aside from the additional money from the Sackler family, the new agreement has one very positive feature. A public document will be created that will include some 33 million documents, such as e-mails, letters and depositions, that go back 20 years. During the course of this litigation, 13 million documents were available to the plaintiffs but not the general public. Maura Healey, attorney general of Massachusetts, predicted that this public repository “will tell the whole story; all the conversations, all the discussions, all the planning, all the ways in which they were going to make money and evade accountability and regulation.”
There have been additional lawsuits filed against other major drug manufacturers (Purdue Pharma was not the only company selling opiate drugs) and major drug distributors. In the past few months, it looks like agreement might be reached in a deal with three of the largest drug distributors and a drug manufacturer. The three drug distributors were McKesson, Cardinal Health and AmerisourceBergen, and the drug manufacturer was Johnson & Johnson. Between them, the three drug distributors had shipped more than three-quarters of the nation’s opioids to pharmacies. They did not alert authorities even when the quantities they were shipping were wildly disproportionate to the local population. However, the drug companies are planning to use tax breaks to offset some of their liability under this plan.
As an example of actions of the three distributors, over a 10-year period these companies “shipped 21 million prescription painkillers to two pharmacies four blocks apart in a West Virginia town with a population of 2,900.” The drug distributors reached this tentative agreement in order to avoid two major trials that had been scheduled for January 2021. I don’t know anything more recent about the deal. It was extremely complicated because it combined lawsuits from both states and municipalities that had suffered from the terrible opioid epidemic (and all of the many plaintiffs had to sign onto the deal or refuse it). The plan calls for the drug distributors to pay $21 billion over a period of 18 years, with McKesson contributing $8 billion of this.
In 2021, the consulting firm McKinsey & Company agreed to a $573 million settlement for advice to Purdue Pharma over marketing their drug OxyContin. As is the case in such settlements, McKinsey will not admit to wrongdoing, but it agreed to court-mandated restrictions on its work with some types of medications. It also agreed to put thousands of pages of documents related to its opioid work into a database that will be accessible to the public.
Even after 2007, when Purdue Frederick had pleaded guilty to felony misbranding of OxyContin, McKinsey continued to advise Purdue Pharma to “focus on selling lucrative high-dose pills,” and to prescribe them for longer periods of time. McKinsey also advised Purdue to “band together” with other companies that marketed opiates, to stop the FDA from “strict treatment” of their product.
This agreement was noteworthy because in the past, consulting firms like McKinsey have been able to escape liability for their recommendations to companies that have either been convicted of wrongdoing, such as Enron, or companies that subsequently went bankrupt, such as Swissair. McKinsey’s global managing partner Kevin Sneader stated that “We deeply regret that we did not adequately acknowledge the tragic consequences of the epidemic enfolding in our communities. With this agreement, we hope to be part of the solution to the opioid crisis in the U.S.”
That was not McKinsey’s reaction in 2018, when Massachusetts sued the company for their role in the opioid crisis. One of the partners in McKinsey’s pharmaceutical line, Martin Elling, wrote to fellow partner Arnab Ghatak, “It probably makes sense to have a quick conversation with the risk committee to see if we should be doing anything” apart from “eliminating all our documents and emails. Suspect not but as things get tougher someone might turn to us.”
On June 26, 2021, Johnson & Johnson agreed to pay $230 million to settle New York lawsuits against it. Johnson & Johnson had developed a new type of poppy called the Norman poppy. It was unique in that it did not contain morphine; however, it had high levels of thebaine, which is a starting ingredient in manufacturing oxycodone and hydrocodone. A J&J subsidiary, Noramco, supplied the bulk of its thebaine to Purdue Pharma, which then used it to produce OxyContin. By the time J&J sold Noramco to a private investment firm in 2016, Noramco was the top producer of opioids in the U.S.
Through its subsidiary Janssen, J&J also marketed Duragesic transdermal fentanyl patches. In 2001, Janssen urged their sales reps to target the top 10,000 opioid prescribers, in an effort to get them to switch to their Duragesic product. In 2003, one sales rep described his lunch with a physician, “Seems a bit nervous about higher doses of [Duragesic] or [OxyContin] … Need to continue to bring up his comfort level for using higher doses of [Duragesic] when necessary.” The McKinsey consulting agency, notorious for their terrible advice to Purdue Pharma, held a series of workshops with J&J in 2002. In order to increase sales, McKinsey suggested “Target high abuse-risk patients (e.g., males under 40).” J&J denied that the company had ever targeted patients at high risk of abuse.
As part of the New York settlement, Johnson & Johnson will permanently stay out of the opioid business in the U.S. New York, collaborating with various counties that had filed separate suits, had accused J&J’s subsidiary Janssen of misleading the public by denying that their drugs were highly addictive, and ignoring warnings of abuse of their products.
In June 2021, a trial was scheduled to begin in New York for several companies involved in the manufacture, distribution, and prescribing of opioids. As we mentioned, Purdue Pharma’s case has been removed from this trial because of their bankruptcy proceedings. Three of the four pharmacy chains originally included in the suit – Walmart, Rite-Aid and CVS – announced that they had reached agreements with New York counties and have been removed from this suit, although Walgreens remains a defendant.
Summary:
The opioid epidemic in America is a major tragedy. As we pointed out in this blog, the central player in this drama was Purdue Pharma. Their product OxyContin turned out to be the equivalent of a nuclear bomb. Here was a product that was twice as potent as morphine, being marketed in doses that reached 80 mg of pure oxycodone (Purdue even marketed for a short time a 160 mg pill).
And Purdue Pharma sales reps encouraged physicians, particularly primary-care doctors who were naïve about opiates, to prescribe OxyContin for conditions such as knee pain, back pain, migraine headaches or fibromyalgia. The Purdue sales reps were carefully coached to urge doctors to prescribe OxyContin for all these relatively minor pains. OxyContin, they said, was the drug “to start with and to stay with.” They reassured hesitant doctors by telling them that the time-release coating of the pill made it impossible for the drug to be abused. The slow, steady release of the oxycodone, they said, prevented the “peaks and troughs” associated with opiates. Furthermore, patients would not experience the “rush” normally associated with opiates, because the pain would offset any high associated with the drug. The sales reps also stressed that addiction was practically unknown for patients taking opiates under the care of a physician. They also pushed the claim that the addiction rate for patients taking opiates from doctors was “well under 1%.”
As it turns out, all of those statements were either false or unproven. And, as OxyContin began to take off as a best-selling drug, it became clear to Purdue Pharma that it was easy to overcome the “time-release” mechanism: the user could simply crush the drug, release the powder inside the coating, and snort or inject it, getting the full power of the opiate at once. In fact, the label of the drug warned that if the pills were crushed, the user would get the full dose of opiate.
Even before OxyContin was released, Purdue Pharma knew that it was easy to abuse the drug, because they knew that users of their earlier time-release morphine pill MS Contin were peeling off the coating and injecting the morphine – in fact, the purple-colored pills had the nickname “purple peelers.” Also, they were aware as early as 1997 that OxyContin was being abused. However, the company insisted that it had no knowledge of any abuse of OxyContin until 2000, when the attorney general of Maine issued a public warning about abuse of their drug.
Purdue Pharma simply ignored the fact that their product was being widely abused; they took the position that the people who abused their product were criminals, and that this was not their problem. For many years they managed to stave off lawsuits while they raked in tremendous profits from their drug, and while an opioid epidemic, fueled by their product, swept the U.S. When it looked like trials were inevitable, they would pay off the defendants, while getting all of the pre-trial evidence sealed from public view.
In 2007, Purdue Pharma was taken to court by John Brownlee, the U.S. attorney in Western Virginia. The company was charged with three felony counts of misbranding, conspiracy and money laundering. Also charged with felonies were three of Purdue’s top executives, including their head legal counsel. However, at the last minute, top officials at the Justice Department overruled Brownlee. Purdue agreed to plead to one count of felony misbranding, while the three executives agreed to plead to misdemeanor misbranding. The company paid a $600 million fine, and the court records and evidence accumulated by the prosecutors was sealed from public view.
By pleading guilty to a federal felony, Purdue Pharma would no longer have been able to do business with government entities such as Medicare and Medicaid. In practice, this would have meant that they could no longer market OxyContin. However, in the final agreement, Purdue Pharma’s parent company Purdue Frederick was allowed to enter the guilty plea. Purdue Frederick then went out of business. However, Purdue Pharma continued to market OxyContin just as before. They changed few of their business practices, but continued to train their sales reps to make assertions about their drug that they knew were false or without corroborating evidence.
At last, the record of lies and deception caught up to Purdue Pharma. In 2020, Purdue Pharma declared bankruptcy, and its assets are being divided up among all of the state and local jurisdictions that are suing the company. The company’s assets (what is left of them, after the Sackler family systematically looted Purdue Pharma’s assets over a 12-year period) will be parceled out to the states, and some of this money will go toward treating the millions of Americans who have suffered through opioid addiction. However, as usual, the company will admit no wrongdoing, no Purdue Pharma executives will see jail time, and the Sackler family that controlled Purdue Pharma will remain one of America’s wealthiest families.
Purdue Pharma executives always complained that plenty of other companies were marketing opioid drugs as well. And they had a point. For example, Vicodin (5 mg hydrocodone bitartrate and 300 mg acetaminophen) contains hydrocodone, and it has also been abused. However, an 80 mg OxyContin tablet contains 16 times the opiate of Vicodin. In addition, in order to obtain a similar “hit” to an 80-mg Oxy pill, users would end up taking so much acetaminophen that they would suffer serious liver damage. Keith Humphreys, a psychology professor at Stanford, described Purdue Pharma’s behavior. “The whole time they’re taking the money, knowing that something is wrong. That is really disgusting.”
We believe that it is fair to say that, in the absence of OxyContin, the American opioid epidemic would be far smaller than we have experienced. In the past twenty years, nearly half a million Americans have died from opioid overdoses. But Purdue Pharma did not cause the opioid epidemic purely on its own. It needed assistance from several other areas.
The first was the physicians and researchers who advocated a new “pain management” paradigm. Their initial claim was that the United States was suffering from an “epidemic of untreated pain.” It is true that many Americans experience chronic pain in varying degrees. And this group of physicians wanted to alleviate this pain. However, many of these new “pain management” gurus claimed that the most effective substances for relieving pain were opiates. Pain “gurus” such as Russell Portenoy, Kathleen Foley, David Joranson and David Haddox pushed hard to remove the “stigma” attached to the use of opiates for pain. These doctors stressed that strong opioid drugs could be used, not merely to relieve pain in the final stages of life for terminally-ill patients, but for those suffering from “non-malignant” chronic pain, for example, back pain. They further argued that under the care of physicians, patients taking opiates would only rarely develop an addiction. The phrase “less than 1% develop addictions” was frequently used, even though there was no hard evidence for this statement. These doctors also asserted that patients would not experience withdrawal symptoms even when the drugs were stopped rapidly – once again, a claim with no strong evidence backing it up.
Furthermore, David Haddox claimed that patients taking opiates who began to show signs of dependence were not experiencing addiction but a phenomenon he called “pseudo-addiction.” The apparent withdrawal symptoms, said Haddox, were simply signs that the patient’s pain was not being alleviated. The cure, said Haddox, was to increase the dosage of opiates. Once the dose was sufficient to relieve the pain, the patient would cease to exhibit withdrawal symptoms. Anyone familiar with opioid addiction would know that progressively increasing the dose of an opiate would often lead to addiction. What was Haddox’s pseudo-addiction based on? Not any robust experimental evidence, but on the basis of a single patient that he had observed. An employee at Purdue Pharma remarked on Haddox’ pseudo-addiction theory, “The solution is just ‘Give them more opioids!’ I don’t think you need a PhD in pharmacology to know that’s wrong.”
The new “pain management” advocates were rewarded handsomely by Purdue Pharma and other pharmaceutical firms. These firms bankrolled “astroturf” organizations such as the American Pain Foundation, the American Academy of Pain Medicine, and the Pain Care Forum. Although these appeared to be independent associations, they were funded primarily by the pharmaceutical companies. Several of these doctors were also handsomely compensated as consultants by companies such as Purdue Pharma, or were hired to conduct “pain management seminars” offered to doctors.
The claims about opioids made by “pain management” advocates were almost all proved false. Anyone with a decent sense of history would realize that opiates as strong as OxyContin should not be used for relatively minor pain. They should be used for the absolute minimum time necessary, and the patient should be taken off strong opiates at the earliest possible date. It would be nice to imagine that doctors such as Russell Portenoy, Kathleen Foley, David Joranson and David Haddox might feel some personal responsibility for their role in fueling the opioid epidemic. We have seen testimony from Portenoy regarding his role in this reign of death – but since Portenoy’s statements were made in return for avoiding prosecution, we might discount those. We have not seen any second thoughts from the other physicians.
We have singled out some of the more outspoken “pain management” advocates for their awful claims. However, the American medical establishment also failed to carry out its responsibilities. During the Obama administration, “White House officials proposed a rule … that would have required mandatory training for doctors prescribing powerful opioids like OxyContin. But lobbyists for the American Medical Association made it clear to administration officials that they would go to war over such a proposal, and the White House backed down.”
The major pharmacy chains were also culpable. They filled millions of prescriptions for “pill mill” doctors, when they had to know that so many opioids were not being prescribed for legitimate purposes. At present major pharmacy chains such as Walgreens, Walmart, CVS and Rite-Aid are being sued for their role in helping to fuel the lethal epidemic. And the major distributors of these drugs also bear responsibility for the epidemic. Barry Meier reports that over the period 2007 to 2012, “the three biggest wholesalers of prescription drugs in the United States – McKesson, Cardinal and AmerisourceBergen – shipped 780,000,000 pain pills containing oxycodone or hydrocodone to West Virginia.”
The opioid epidemic could also have been largely averted had the U.S. government regulatory agencies done their job. OxyContin had to be approved by the FDA; unfortunately, Curtis Wright, the FDA official personally responsible for approving the drug, failed to require that Purdue Pharma prove that their drug could not be abused. He also allowed unusual wording on the package insert: “Delayed absorption, as provided by OxyContin tablets, is believed to reduce the abuse liability of the drug.” This remarkable unproved assertion was used by Purdue Pharma to justify their claims that the drug could not be abused – which turned out to be tragically false. Wright could have forced Purdue to carry out extensive tests to prove the drug could not lead to addiction; he did not. After approving OxyContin Wright resigned from the FDA; one year later he was hired by Purdue Pharma, a company he had contacted about possible employment even before he left the FDA.
One of Purdue Pharma’s tactics was to hire many of its own critics. Jay McCloskey was the top federal prosecutor in Maine. In 2000, he issued a letter to doctors in Maine warning them of rampant abuse of OxyContin and other opioid drugs in his state. Purdue Pharma subsequently hired McCloskey, who later testified that he was “deeply impressed by the unmistakable interest in the public welfare” shown by his new employer. In 2001, U.S. Attorney for Eastern Kentucky Joe Famularo called OxyContin a “locust plague” infecting Kentucky. But after Purdue Pharma brought him in as a consultant Famularo asserted that he now considered OxyContin “a fine product.”
In addition to its own legal team, Purdue Pharma utilized a team of “hired gun” lawyers. They particularly focused on people with prior experience in government. Howard Shapiro had earlier been general counsel to the FBI; his firm was paid over $50 million for their work on behalf of Purdue Pharma. Eric Holder had been an assistant attorney general in the Clinton administration. Mary Jo White had been the U.S. Attorney for the Southern District of New York and was later appointed chair of the Securities and Exchange Commission. She has represented Purdue Pharma and members of the Sackler family since 2005. Rudy Giuliani began working as a consultant after serving as mayor of New York. These lawyers used both their legal expertise and their political connections to defend Purdue Pharma in numerous lawsuits; they also worked hard to shield members of the Sackler family from any culpability for their actions.
During the time that these lawyers were defending Purdue Pharma, hundreds of thousands of Americans became addicted to opiates and died. We wonder if any of these lawyers feel any responsibility for allowing Purdue Pharma and the Sackler family to continue making obscene profits from a drug that was being dishonestly marketed and was fueling this horrible American epidemic of drug abuse and death. At least, one might hope that their law firms would direct some of their Purdue Pharma legal fees towards addiction treatment programs.
We are now winding through a major round of class-action lawsuits relating to the opioid epidemic. A colleague describes the typical class-action suit as one where “lawyers make $200 million, while each individual affected gets $3.16.” The end-game is a predictable one: several companies will pay large sums of money, but will admit to no wrongdoing. And despite breaking the law and causing untold damage, no corporate executives will do any jail time. White-collar crime is generally deliberate, and systematically planned by groups of people. For example, recently Volkswagen illegally created a system to cheat emissions testing with their diesel cars. Clearly, this tactic was known of and approved by VW engineers, top executives and lawyers. If white-collar criminals were sentenced to jail time, it would seem that this would be a significant deterrent to future white-collar crime. Top corporate executives might be loath to sign off on an illegal scheme if they could serve jail time for their actions.
It appears that finally, the medical community understands what should have been glaringly obvious from the start: powerful opiates such as 80 mg OxyContin should not be used for minor chronic pain. They should be drugs of last resort. If they are prescribed, it should be at the smallest possible dose for the shortest possible time. We hope that government entities such as the Food and Drug Administration will step up to the plate and prevent their drug-approval process from being co-opted by bad actors such as Purdue Pharma. And we hope that the money obtained from opiate class-action lawsuits will be directed towards treatment programs for those areas that have been impacted most by America’s terrible opioid epidemic.
Source Material:
Sam Quinones, Dreamland: The True Tale of America’s Opiate Epidemic, Bloomsbury Publishers, 2015
Patrick Radden Keefe, Empire of Pain: The Secret History of the Sackler Dynasty, Doubleday 2021;
Patrick Radden Keefe, The Family That Built an Empire of Pain, The New Yorker, Oct. 23, 2017
History.com, Heroin, Morphine and Opiates:
National Institute on Drug Abuse: Heroin Drug Facts,
Wikipedia, Addiction Rare in Patients Treated With Narcotics
M.S. Green and R.A. Chambers, Pseudo-Addiction: Fact or Fiction? An Investigation of the Medical Literature, Current Addiction Reports 2, 310 (2015)
Barry Meier, Pain Killer: An Empire of Deceit and the Origin of America’s Opioid Epidemic. Random House, 2018.
Francis X. Clines and Barry Meier, Cancer Painkillers Pose New Abuse Threat, New York Times, Feb. 9, 2001.
Barry Meier, Origins of an Epidemic: Purdue Pharma Knew Its Opioids Were Widely Abused, New York Times, May 29, 2018
Barry Meier, Every Time I Thought the Purdue Pharma OxyContin Story Was Over, I Was Wrong, New York Times, June 8, 2018
Russell Portenoy and Kathleen Foley, Chronic Use of Opioid Analgesics in Non-Malignant Pain: Report of 38 Cases, Pain 25, 171 (1986).
J. Porter and H. Jick, Addiction Rare in Patients Treated With Narcotics, New England Journal of Medicine 302, 123 (1980)
Jose L. Medina and Seymour Diamond, Drug Dependency in Patients With Chronic Headaches, Headache, March 1977;
Samuel Perry and George Heidrich, Management of Pain During Debridement; A Survey of U.S. Burn Units, Pain 13, 267 (1982)
FBI, Sept 2012, Bloomington Man Sentenced On Health Care Fraud, Drug Distribution Charges,
Vic Rykaert, Board Suspends Dr. Tristan Stonger’s License in Drug Investigation, Indy Star Apr. 15, 2016,
David E. Weissman and David J. Haddox, Opioid Pseudoaddiction – an Iatrogenic Syndrome, Pain 36, 363 (1989).
Thomas Catan and Evan Perez, A Pain-Drug Champion Has Second Thoughts, Wall Street Journal Dec. 17, 2012.
Caitlin Esch, How One Sentence Helped Set Off the Opioid Crisis, Marketplace Dec. 13, 2017
Harriet Ryan, Lisa Girion and Scott Glover, ‘You want a description of hell?’ OxyContin’s 12-hour problem, Los Angeles Times, May 5, 2016
Harriet Ryan, Lisa Girion and Scott Glover, More Than 1 Million OxyContin Pills Ended Up in the Hands of Criminals and Addicts. What the Drugmaker Knew, Los Angeles Times, July 10, 2016.
Harriet Ryan, Lisa Girion and Scott Glover, OxyContin Goes Global — “We’re Only Just Getting Started,” Los Angeles Times Dec. 18, 2016
Michael Forsythe and Walt Bogdanich, McKinsey Settles for Nearly $600 Million Over Role in Opioid Crisis, New York Times Feb 3, 2021.
N. Evans, E. Lieber and N. Power, How the Reformulation of OxyContin Ignited the Heroin Epidemic, NBER Working Paper 24475, April 2018.
Christopher Glazek, The Secretive Family Making Billions From the Opioid Crisis, Esquire, Oct 16, 2017.
Patrick Radden Keefe, The Sackler Family’s Plan to Keep its Billions, They New Yorker, Oct 4, 2020
Bethany McLean, “We Didn’t Cause the Crisis:” David Sackler Pleads His Case on the Opioid Epidemic, Vanity Fair, June 19, 2019
Dept of Justice release on Purdue Pharma, Nov 24, 2020
Ian Hoffman, $26 Billion Settlement Offer in Opioid Lawsuits Gains Wide Support, New York Times Nov. 6, 2020
Drug Companies Plan Tax Breaks to Offset $26 Billion Opioid Settlement, NPR News Mar 9, 2021
Julia Lurie, Inside Johnson & Johnson’s Quiet Domination of the Opioid Market, Mother Jones, June 11, 2019
Sarah Maslin Nir, Johnson & Johnson to Pay New York $230 Million to Settle Opioid Case, New York Times, June 26, 2021
Jan Hoffman, 15 States Reach a Deal With Purdue Pharma, Advancing a $4.5 Billion Opioids Settlement, New York Times, July 8 2021 https://www.nytimes.com/2021/07/08/health/purdue-pharma-opioids-settlement.html
