Note added on September 18, 2023: We have been alerted by a reader to the site DrugWatch, which provides consumers with information about potential dangers and side-effects of medicines and chemicals in wide usage. Two specific pages on that site — Glyphosate and Cancer and Understanding Glyphosate — provide useful information about the chemical glyphosate, a main ingredient in the weed killer Roundup, which we treat as one of the two case studies in our post below.
July 3, 2020
There are hundreds of sagas exhibiting the roles of the product defense network and techniques we described in Part I of this post, in denying or postponing labeling and regulation of toxic products as health hazards, and lawsuits seeking recompense for damages suffered. In this part, we will describe two recent and ongoing case studies in some detail. These are illustrative of the sometimes decades-long product defense efforts and the harm done along the way to workers, consumers and unlucky citizens who live near production factories. Many additional cases are outlined in David Michaels’ book The Triumph of Doubt and in the Center for Public Integrity story Meet the ‘Rented White Coats’ Who Defend Toxic Chemicals.
I. The Forever Chemicals
The story of per- and poly-fluoroalkyl substances, or PFAS, is a quintessential tale of toxic chemicals: accidental discovery of substances with magical properties, leading to enormous profits for the producers; early indications of toxicity long hidden from consumers and regulators; a lengthy unraveling of the harmful impacts, led by a lone legal crusader, whose story has been told in a 2016 New York Times Magazine article and in the 2019 film (“Dark Waters”) based on it; decades of efforts by product defenders to contradict and downplay the epidemiological science of the health hazards; large eventual monetary settlements to multiple lawsuits, which nonetheless were dwarfed by the profits the producers reaped over the years; ongoing efforts by the U.S. federal government to downplay the continuing exposure risk to many millions of citizens.
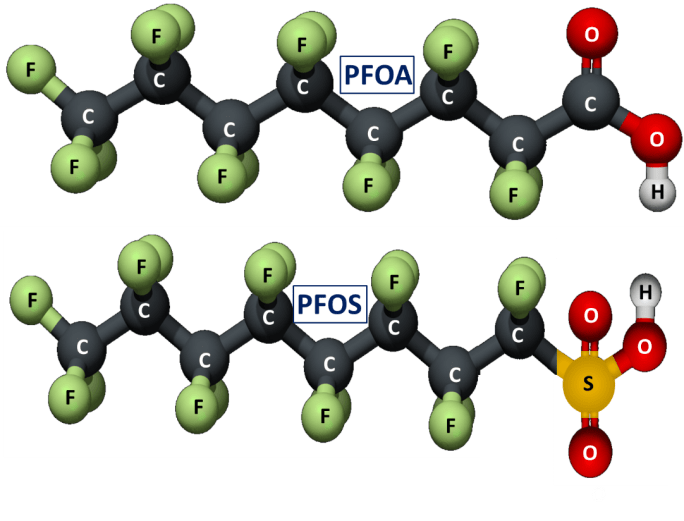
PFAS refer to a large class of man-made fluorocarbon chemicals, of which the two most prominent are illustrated schematically in Fig. 1. PFOA was used by DuPont to produce Teflon, the non-stick material first accidentally synthesized in a DuPont laboratory in 1938. PFOS was first developed in association with the Manhattan Project during World War II. After the war, the 3M company purchased the patent, and discovered accidentally in 1953 that it could be used to create a product that repelled water and grease, a stain-resistant product later patented as Scotchgard. PFAS molecules are extremely stable because the chemical bonds are hard to break. Along with their other properties, that stability has led to the wide array of products that now utilize PFAS, as illustrated in Fig. 2. But that stability also makes the PFAS chemicals unusually resistant to breakdown in the environment, in water systems, and in human bodies, in all of which they accumulate over time. Hence, they are sometimes referred to as the “forever chemicals.” They are also ubiquitous worldwide.

Beginning in 1951, PFOA was produced in the U.S. only by the 3M company, and was purchased from them by DuPont to prevent clumping of material during the fabrication of Teflon. Though the health hazards of PFOA were not yet known, 3M had recommended to DuPont that they dispose of it via incineration or chemical waste facilities. In 1998, 3M scientists launched a program to look for PFAS contamination in human blood samples from multiple worldwide populations. It was found to be present at detectable levels for all the studied population samples, except for one from the late 1940s, before PFOA had been widely used in commercial products. Under pressure from the EPA, 3M announced a phaseout of its production of PFOA and PFOS, and hence of its lucrative Scotchgard product, in 2000. But because the chemicals last “forever,” 3M was hardly off the hook for later lawsuits regarding its legacy disposals of the chemicals. But DuPont was hardly ready to give up on Teflon. In 2002, it built its own PFOA production plant in Fayetteville, North Carolina.
Public awareness of the health hazards of PFAS began to emerge at the turn of the 21st century, after farmer Wilbur Tennant, whose land was adjacent to a landfill for waste from the DuPont Washington Works factory in Washington, West Virginia, noticed his cows contracting strange diseases and dying in large numbers. Tennant suspected toxic runoff from the DuPont landfill to be the source of the problem, but had no success getting local help on the issue, since the factory was the major employer around Parkersburg. His neighbors suggested he contact Rob Bilott (see Fig. 3), who had visited their farm often as a young boy, and who was then a corporate lawyer for a large firm in Cincinnati. Bilott specialized in helping chemical corporations deal with environmental regulations but, after seeing the video evidence Tennant brought to a meeting with him, decided to work against one of the largest of the chemical companies by taking on the farmer’s case. Asked about his decision, Bilott said, “It was a great opportunity to use my background for people who really needed it.’’

Needless to say, DuPont did not take kindly to the suit Bilott filed against them in 1999 in the Southern District of West Virginia. DuPont’s first action was to commission a study of Tennant’s property and cows, carried out by a team of six veterinarians chosen by DuPont and by the EPA. Their report found no culpability on DuPont’s part, but rather attributed the cows’ diseases and death to “poor husbandry.” Bilott had little luck trying to understand the real cause of the problem, until he came across a communication between DuPont and EPA that mentioned PFOA, a chemical Bilott had never heard of, despite his work on environmental law for chemical corporations. In preparation for the trial, he asked DuPont to share all the documentation they had about PFOA, but the company refused, until a court ordered them to do so in the Fall of 2000. In response to the court order, DuPont tried to inundate Bilott by sending him 110,000 pages of information. DuPont’s lawyers must have figured that would end Bilott’s crusade, but instead they learned why toxic producers fear the discovery process in civil lawsuits.
Of his painstaking work reading through the voluminous, historic documentation, Bilott said: “It was one of those things where you can’t believe you’re reading what you’re reading. That it’s actually been put in writing. It was the kind of stuff you always heard about happening but you never thought you’d see written down.’’ The documents revealed that over the years, DuPont had pumped hundreds of thousands of pounds of PFOA powder through outfall pipes at the Washington Works plant into the Ohio River, and dumped an additional 7100 tons of PFOA-laced sludge into open, unlined pits on the property. PFOA thus entered the drinking water supply of some 100,000 inhabitants of surrounding communities.
Even more damaging was the revelation from the documents that DuPont and 3M had been carrying out secret research on the health impacts of PFOA for decades. They had discovered that it caused liver damage, birth defects and cancerous testicular, pancreatic and liver tumors in laboratory animals and that it was retained at high concentrations in the blood of factory workers at the Washington facility. Two of seven tested pregnant employees of DuPont’s Teflon division gave birth to infants with eye defects. Dust vented from chimneys in the factory was found to settle far beyond the property line, and PFOA was found present in the local water supply. In 1991 DuPont scientists had established an internal safety limit of one part per billion PFOA contamination in drinking water available to their employees. They had even found extraordinarily high PFOA concentrations in the creek that ran by their landfill and Tennant’s farm. In 1993, DuPont management had decided against switching to a less toxic alternative to PFOA their scientists had been working on, because it would introduce too much risk into what was then a billion dollar profit per year product. None of this information had been divulged publicly, in their commissioned report about Tennant’s cows, or to the EPA.
When Bilott informed DuPont’s lawyer that he had all of this information, the company quickly settled the Tennants’ lawsuit. But that did not satisfy Bilott, who was concerned about the health of all the people exposed to that drinking water supply. In August 2001, he filed a class action suit against DuPont on behalf of 70,000 residents in six water districts in Ohio and West Virginia, whose drinking water supply had PFOA contamination above the internal company safety limit set by DuPont. In March 2001 he sent a 972-page letter detailing the threat to the Director of every relevant regulatory agency, including EPA, and to the U.S. Attorney General, demanding immediate action to regulate PFOA and clean the local drinking water around Parkersburg.
For the first time, DuPont recognized a formidable adversary, describing Bilott in an internal memo as a threat to “the entire fluoropolymers industry.” They first tried, but failed, to get a court to issue a gag order blocking Bilott from communicating with federal agencies. In light of the files Bilott sent them, EPA launched a health risk assessment process for PFOA, and then filed suit against DuPont in 2004 for violating the 1976 Toxic Substances Control Act. According to that act, the EPA can test chemicals only when it has been provided evidence of harm, but companies are required to inform EPA when they discover toxic effects. DuPont had clearly failed to meet that requirement.
In the face of the class action suit and the EPA assessment, DuPont engaged the product defense industry in full force. They first recommended to the state government of West Virginia that the product defense firm TERA, under the leadership of Michael Dourson, could assemble an “independent” panel of experts to advise the state as to the safe level for humans of PFOA contamination in drinking water. According to an internal DuPont e-mail uncovered in subsequent litigation, TERA was chosen because they could be trusted to “assemble a package and then sell this to EPA, or whomever we desired.” And Dourson came through, convincing West Virginia to adopt a safety limit of 150 ppb in 2002, when DuPont’s own internal safety limit was 1 ppb and toxicologists advising Bilott were recommending 0.2 ppb. On what basis the TERA panel determined this limit we do not know, because the West Virginia Department of Environmental Protection – with several lawyers regularly used by DuPont then in leadership positions – destroyed the evidence as part of their “standard practice and policy of destroying documents they anticipate might be the subject of a subpoena in this litigation.”
In addition, DuPont hired ChemRisk to downplay the risk to the plaintiffs in Bilott’s class action suit. ChemRisk’s Dennis Paustenbach argued that his estimate of the daily intake of PFOA by residents living within five miles of the Washington Works plant over the preceding 50 years would have been 10,000 times below the safe limit established by an “independent” panel – that would be the TERA panel that DuPont helped to set up.
In 2003, DuPont further engaged the Weinberg Group to help them navigate the legal and regulatory threats. In a remarkable April 2003 letter, the Weinberg Group’s vice president for product defense, P. Terrence Gaffney, laid out their strategy in detail for DuPont vice president Jane Brooks, thereby providing a discoverable written “blueprint” of the allegedly “independent” pseudo-scientific approach of the product defense industry. Here are some excerpts from that letter:
“We must implement a strategy at the outset which discourages governmental agencies, the plaintiffs’ bar, and misguided environmental groups from pursuing this matter any further than the current risk assessment contemplated by the Environmental Protection Agency (EPA) and the matter pending in West Virginia…We can assist with all phases of the technical and scientific defense, but more importantly, shape the debate and direction of the PFOA issue… we will harness, focus, and involve the scientific and intellectual capital of our company with one goal in mind—creating the outcome our client desires…This would include facilitating the publication of papers and articles dispelling the alleged nexus between PFOA and teratogenicity as well as other claimed harm… it matters little that the industry is satisfied PFOA is safe. The real issue is the perception outside the industry. This battle must be won in the minds of the regulators, judges, potential jurors, and the plaintiffs’ bar…Ours is a task-oriented organization in which clients make specific assignments under carefully planned, client-controlled budgets… some preliminary suggestions of tasks for managing issue related to PFOA include:
-
- develop ‘blue ribbon panels’ of thought leaders on issues related to PFOA IN REGIONS WHERE MANUFACTURING PLANTS ARE LOCATED to create awareness of safety regarding PFOA in areas of likely litigation, and in particular where medical monitoring claims may be brought;
- develop an aggressive campaign focused on the safety and utility of PFOA and the products in which it is used;
- coordinate the retrieval, organization, and analysis of literature to date (both internal and extemal) regarding safety of PFOA and create a centralized searchable database for industry use;
- begin to identify and retain leading scientists to consult on the range of issues involving PFOA so as to develop a premium expert panel and concurrently conflict out experts from consulting with plaintiffs;
- begin to coordinate focus groups of mock jurors to determine the best ‘themes’ for defense verdicts and perspectives on management of company documents and company conduct;
- reshape the debate by identifying the likely known health benefits of PFOA exposure by analyzing existing data, and/or constructing a study to establish not only that PFOA is safe over a range of serum concentration levels, but that it offers real health benefits (oxygen carrying capacity and prevention of CAD);
- coordinate the publishing of white papers on PFOA, junk science and the limits of medical monitoring;
- work with industry lobbyists to ensure they remain on message regarding the scientific issues related to PFOA;
- provide the strategy to illustrate how epidemiological association has little or nothing to do with individual causation.”
By 2002, the EPA issued preliminary findings that PFOA might indeed pose human health risks not only to those drinking tainted water, but also to those who cooked with Teflon pans and to members of the general public. By 2003, it was revealed that PFOA appeared in blood samples of adult Americans at an average concentration of 4—5 ppb. In the light of these developments, and in spite of the efforts of their product defense teams, DuPont chose to settle the lawsuits. They got off easy with the EPA during the George W. Bush administration, paying a $16.5 million fine in 2005, without having to admit liability. This was the largest civil penalty the EPA had ever obtained up to that point, but it was a pittance for DuPont.
The September 2004 settlement of the class action suit was not so straightforward. Bilott had brought the suit under a recent West Virginia state law that recognized “medical-monitoring” claims. According to this law, a plaintiff needed only to prove that he/she had been exposed to a toxin, without the necessity of proving that a subsequent medical condition was a direct result of that exposure. If the plaintiff were to win such a case, then the defendant would be required to fund regular medical tests of the plaintiff. If the plaintiff then later fell ill, he/she would be entitled to sue retroactively for damages. In the settlement, DuPont agreed to pay a cash award of $70 million to the 70,000 plaintiffs, plus additional amounts to improve the water treatment facilities involved and to fund a research program to determine whether there was a “probable link” between PFOA exposure and any diseases. If such links were found to exist, then DuPont would pay for medical monitoring of the plaintiffs in perpetuity. Until that research program delivered results, plaintiffs were forbidden from filing personal injury suits against DuPont.
Bilott outsmarted DuPont again. He got the class action plaintiffs to agree to provide their medical information and to donate their blood samples for the research program, in exchange for receiving their share of the settlement payout. The epidemiologists working on the research program then had inexhaustible funding from DuPont, as part of the settlement, plus medical records and blood samples from 69,000 plaintiffs. This fueled the largest epidemiological study in history. Because there was so much data and so many potential disease links to explore, it took a full seven years (and cumulative $33 million of DuPont funding) before the program produced publicly available results.
During that interval – an excruciating delay for the plaintiffs and a time of enormous stress for Bilott – DuPont and the chemical industry front organizations kept up the public relations claiming that there were negligible risks to human health. The misleadingly titled American Council on Science and Health reported in 2005 that “the current data indicate that we can expect no risk to human health associated with the levels of PFOA exposure found in the general population.” But by the end of 2011, the exhaustive study resulting from Bilott’s class action suit had established “a ‘probable link’ between PFOA and kidney cancer, testicular cancer, thyroid disease, high cholesterol, pre-eclampsia and ulcerative colitis,” while finding no evidence to support a possible link to a variety of other conditions.
Armed with these findings, Bilott worked with more than 3500 former plaintiffs who had suffered from the linked diseases to file individual personal injury lawsuits against DuPont. The first three of these suits that went to court ended with DuPont being hit with a cumulative $19.7M in damages. Meanwhile, DuPont agreed with EPA to cease production of PFOA in 2013. They spun off their chemical businesses to a new corporation called Chemours, and Chemours has been producing the alternative “short-chain” PFAS (named GenX) considered by DuPont scientists back in 1993, but then rejected by DuPont management. In 2017 DuPont and Chemours decided to settle the remaining personal injury lawsuits for a total of $671.7M.
The 2017 settlement may have seemed to end the PFOA saga, but what about all the other chemicals in the PFAS class, including 3M’s PFOS and Chemours’ new GenX? In 2012, Harvard epidemiologist Philippe Grandjean published a study linking PFAS exposure to disruption of immune system function, especially affecting children’s antibody responses to childhood vaccinations. 3M scientists criticized the Grandjean paper, claiming that it was refuted by previous studies. Nonetheless, the National Toxicology Program reviewed the literature spanning both human and animal evidence and concluded in 2016 that “the PFOA used in Teflon and PFOS in Scotchgard are presumed to be immune hazards to humans.” That same year, EPA published a health advisory suggesting that the safe limit of PFOA and PFOS in drinking water should be set at 0.07 ppb, a factor of 2,000 below the level TERA had set for West Virginia in 2002. Grandjean later uncovered evidence that internal 3M research had revealed the impact of PFAS on immune system function back in 1978, but that the evidence had been suppressed by 3M.
Because PFAS don’t break down in the environment, the fact that 3M had ceased producing PFOA and PFOS by 2002 did not shield them from litigation. The state of Minnesota sued 3M in 2016, accusing the local company of dumping large amounts of PFAS waste over many years, all the while knowing, but hiding, the fact that these chemicals were extremely hazardous. 3M lawyers used the same approach DuPont had tried previously, engaging the product defense consultants at Gradient and Exponent to downplay the risk. Gradient scientist Barbara Beck provided a report for the court, claiming that the state overestimated both the exposures and the risk to the Minnesota public. Exponent scientists published a paper in the preferred industry-friendly journal Critical Reviews in Toxicology, reaching the de rigueur conclusion that the “available evidence is insufficient to conclude that a causal relationship has been established between PFOA or PFOS exposure and any immune condition in humans.”
But in the end, 3M settled the Minnesota lawsuit in 2018 for $850M, of course without admitting any liability. But after the settlement, the Minnesota Attorney General publicly released many of the documents she would have used in trial, revealing among many other facts the millions of dollars 3M had paid to academic scientists to produce papers intended as “defensive barriers to litigation.”
And Rob Bilott was not done yet. In 2018 he filed a new lawsuit against several companies, including 3M, DuPont and Chemours, initially on behalf of a firefighter who had used fire-suppressing foams and firefighting equipment utilizing PFAS for 40 years. Bilott’s intention is to make this a class action suit “seeking new studies and testing of the larger group of PFAS chemicals on behalf of a proposed nationwide class of everyone in the United States who has PFAS chemicals in their blood.” That class may include a large fraction of the U.S. population!
The Agency for Toxic Substances and Disease Registry (ATSDR) within the U.S. Centers for Disease Control produced a 2018 report recommending even more stringent minimal risk levels for PFAS contamination in drinking water than were contained in the EPA’s 2016 health advisory. The Trump administration tried to block public release of this report, but capitulated under intense Congressional pushback. The pushback was led by legislators representing districts containing military bases, which had used PFAS-based fire-fighting foams for decades. Also in 2018, a new misleadingly named industry front organization, the Responsible Science Policy Coalition, was formed with funding from 3M and other companies for the express purpose of helping western states avoid lawsuits regarding PFAS contamination of drinking water.
Furthermore, ProPublica reports that two new analyses of drinking water reveal that the EPA and the Department of Defense have for years been using tests designed to downplay PFAS exposure levels, by factors of more than 20. The Environmental Working Group, a public advocacy organization whose scientists have studied PFAS pollution, has estimated that as many as 110 million Americans are now at risk of exposure to dangerous levels of PFAS contamination of drinking water. Their estimate is backed up by detailed maps of the locations in the U.S. where industrial and military site discharges of PFAS are known to have occurred and where drinking water contamination has been measured to exceed the ATSDR minimal risk levels. The mechanisms by which PFAS leaches into our water supply are summarized in Fig. 4.

While many state Departments of Environmental Protection are now grappling with how to clean up their water supply and setting their own safe levels of PFAS contamination, the U.S. EPA has yet to set any enforceable national standard for PFAS contamination. Their website contains the following, rather generic statements:
“There is evidence that exposure to PFAS can lead to adverse health outcomes in humans. If humans, or animals, ingest PFAS (by eating or drinking food or water that contain PFAS), the PFAS are absorbed, and can accumulate in the body. PFAS stay in the human body for long periods of time. As a result, as people get exposed to PFAS from different sources over time, the level of PFAS in their bodies may increase to the point where they suffer from adverse health effects.
Studies indicate that PFOA and PFOS can cause reproductive and developmental, liver and kidney, and immunological effects in laboratory animals. Both chemicals have caused tumors in animal studies. The most consistent findings from human epidemiology studies are increased cholesterol levels among exposed populations, with more limited findings related to:
-
- infant birth weights,
- effects on the immune system,
- cancer (for PFOA), and
- thyroid hormone disruption (for PFOS).”
Finally bowing to pressure from Congress and the 2018 ATSDR report, the EPA published in 2019 a PFAS Action Plan, stating that “the EPA will make a final determination for PFOA and PFOS, and as appropriate, other PFAS and take the appropriate next regulatory steps under the SDWA [Safe Drinking Water Act].” Early in 2020, the Agency issued an Announcement of Regulatory Determinations for Contaminants on the Fourth Drinking Water Contaminant Candidate List, to include PFOA and PFOS, with a comment period just recently closed.
Undoubtedly, a number of the nearly 12,000 comments received came from product defense scientists and supporters, arguing that the preliminary standards EPA was considering are too stringent. For example, the industry front groups American Chemistry Council and Responsible Science Policy Coalition submitted comments of 14 and 20 pages, respectively. The former argues that “although exposure to PFOA and PFOS, at sufficiently high doses, may be associated with health effects in animals,” available model studies to extrapolate to human impacts suggest “that EPA’s current assessment… overestimates the risks associated with low-level exposure to PFOA and PFOS in drinking water.” It will likely take years for the rulemaking process to complete, it is unlikely to address chemicals other than PFOA and PFOS specifically, and it may run into more litigation before it can be implemented.
So more than two decades on, there is an outside chance that Rob Bilott may finally get to see some level of the regulation he demanded in his 972-page letter of 2001. In the meantime, he continues to fight for the victims of these toxic chemicals. The PFAS saga shows that lawsuits are currently a more effective means to combat very real toxic product health impacts than federal agency regulations.
II. Roundup Ready or Not
The scientists at federal regulatory agencies generally have their hands full as they try over years to carry out unbiased evaluations of product health hazards, in the face of the steady onslaught of vociferous pressure from industry lobbyists and their political allies, and the burgeoning pseudoscience for hire produced by product defense teams. But matters are often made even more complicated by the revolving door between industry and regulatory leadership positions. Our next case study reveals the roadblocks set up by collusion between the regulated industries and their friends in high places, over and above product defense efforts analogous to those in the PFAS saga.
The chemical in question is glyphosate (see Fig. 5), the main ingredient of Monsanto’s Roundup weed-killer, which was introduced to the market in 1974. In the years since Monsanto’s patent expired in 2000, it has also been used with different formulations in herbicides developed by other large agri-chemical corporations. Glyphosate use has grown extremely rapidly since the introduction of genetically modified crops in the 1990s. Monsanto and later other agribusinesses have developed and sold genetically modified seeds that produce Roundup Ready crops, which are themselves resistant to the herbicide.
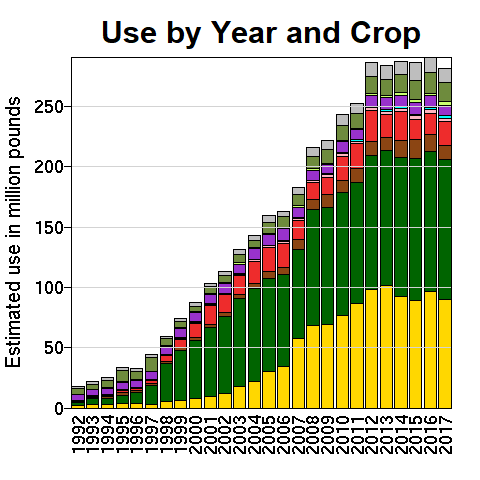

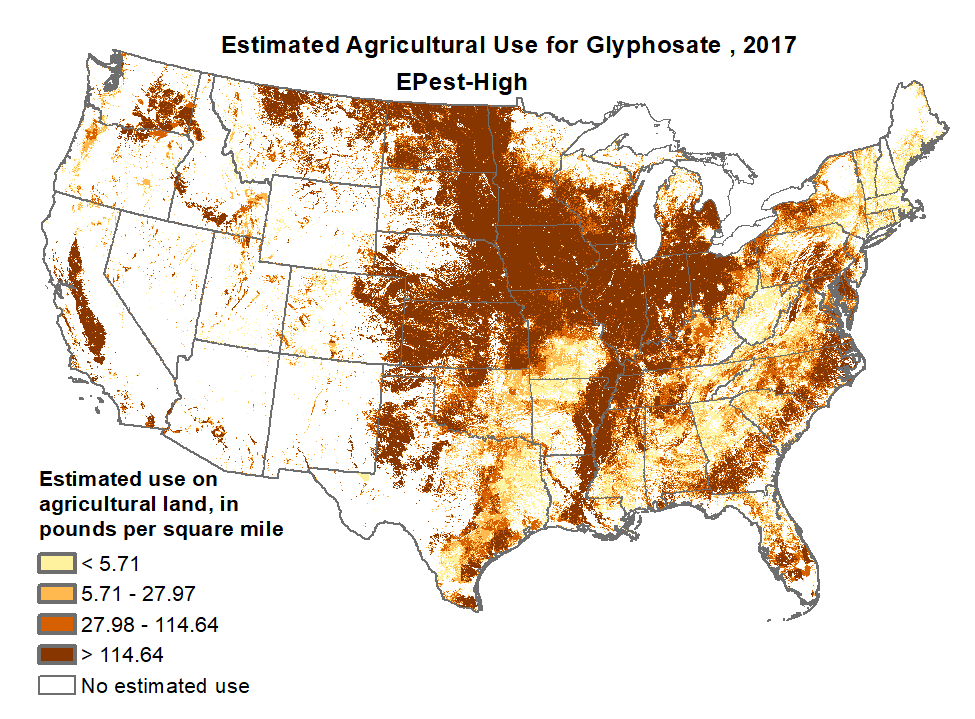
As seen in Fig. 6, the introduction of Roundup Ready crops, especially for corn, soybeans and cotton, have expanded glyphosate usage in the U.S. alone from about 15 million pounds per year in 1992 to a present level around 300 million pounds per year. Glyphosate is by now the most heavily used agricultural chemical in history. As seen in the map in Fig. 7, glyphosate usage is especially prevalent throughout the U.S. midwest and in the agricultural region of California’s San Joaquin Valley. In 2015, 89% of corn, 94% of soybeans, and 89% of cotton produced in the United States were from strains that were genetically modified to be herbicide-tolerant. Glyphosate has been celebrated as a “one in a 100-year discovery that is as important for reliable global food production as penicillin is for battling disease.”
In 1985 an EPA committee determined that Roundup might cause cancer, based in part on an early study in which a few mice exposed to a heavy dose of the herbicide developed a very rare form of kidney cancer. Six years later, after former Monsanto lawyer Michael Taylor began working at the FDA, the EPA reversed its decision, discounting the significance of the mouse study. Given the growing usage of glyphosate seen in Fig. 6 and growing public concerns about GMO crops, it was inevitable that the possible toxicity of glyphosate and commercial formulations based on it would come up for renewed consideration by oversight agencies worldwide. And given that the herbicide sales had grown to a $10 billion per year worldwide industry, it was equally inevitable that the producers would pull out the product defense stops to prevent toxic labeling and possible regulation (see Fig. 8).

Many of the ugly, behind-the-scenes details of Monsanto’s role in these battles have come to light, as in the PFAS case, via documents shared through discovery in multi-plaintiff lawsuits and through Freedom of Information Act releases from federal agencies. And those details have been reported in depth in Carey Gillam’s excellent 2017 book Whitewash: The Story of a Weed Killer, Cancer, and the Corruption of Science and, surprisingly, in a 2018 Congressional report prepared by the minority members of the U.S. House of Representatives Committee on Science, Space and Technology.
The issue with potential toxic labeling of glyphosate came to a head with a 2014 announcement by the World Health Organization’s International Agency for Research on Cancer (IARC) that it was convening a panel of experts to review published evidence regarding links of cancer to a group of pesticides, including glyphosate. In the U.S. pesticide regulation is provided by the EPA, and Monsanto felt they had considerable sway over EPA decisions. For example, in 2013 the EPA had just acceded to Monsanto’s request to raise “the legally allowed amount of glyphosate residues in food considered safe to levels far higher than in other countries.” But Monsanto was seriously worried about the independence of the IARC panel, which was to include no industry scientists and to exclude unpublished studies, such as the ones chemical companies often carry out to get regulatory approval.
Furthermore, as would later be revealed in discovery documents, Monsanto had been aware of toxic vulnerabilities for Roundup for well over a decade, though that did not stop them from marketing Roundup as a product “as safe as table salt.” In 1999 they had funded geneticist James Parry of Swansea University in Wales to evaluate glyphosate’s genotoxicity (i.e., capability of causing DNA damage that might lead to cancer). They then worked successfully to prevent Parry from publishing his results, which had shown that “glyphosate is capable of producing genotoxicity both in vivo and in vitro by a mechanism based upon the production of oxidative damage.” Monsanto scientist William Heydens urged in an e-mail that Monsanto identify alternative genotoxicity experts to Parry, because Monsanto is “currently very vulnerable in this area…We want to find/develop someone who is comfortable with the genetox profile of glyphosate/Roundup and who can be influential with regulators and scientific outreach operations when genetox issues arise.” In 2001, arguing against helping Parry with further research, Heydens said in an e-mail “Data generated by academics has always been a major concern for us in the defense of our products” (see Fig. 8).
However, Monsanto did find a few academic scientists who were willing to be fronts for the corporation. One was Bruce Chassy, a Professor and former Head of the Department of Food Science and Human Nutrition at the University of Illinois, and a former Monsanto grantee. In 2010 he colluded with Monsanto to launch a putatively independent non-profit website Academics Review, as “an association of academic professors, researchers, teachers and credentialed authors from around the world who are committed to the unsurpassed value of the peer review in establishing sound science. We stand against falsehoods, half-baked assertions and theories or claims not subjected to this kind of rigorous review.” Chassy may have wanted to combat pseudoscience opposing genetic engineering of crops, but he allowed himself to be co-opted into defending Roundup. In a November 2010 e-mail to Chassy, Monsanto Science and Policy lead Eric Sachs gave up the game: “You and I need to talk more about the ‘Academics Review’ site and concept…From my perspective, the problem is one of expert engagement and that could be solved by paying experts to provide responses…The key will be keeping Monsanto in the background so as not to harm the credibility of the information.”
Sachs recruited Chassy again when Monsanto needed help combating an independent 2012 article by French microbiologist and GMO opponent Gilles-Eric Séralini, which appeared in the journal Food and Chemical Toxicology (FCT). The research reported in that article found that Roundup and Roundup Ready corn caused tumors in rats. FCT Editor in Chief for Vision and Strategy Wally Hayes – a Professor at the Harvard School of Public Health who had signed a consulting agreement with Monsanto a month earlier — alerted Monsanto to the imminent publication of this article in the journal and said he needed some Letters to the Editor to cast doubt on Séralini’s work. Chassy was one of the scientists Monsanto recruited to write such letters, although Sachs told his Monsanto colleagues “We should not provide ammunition for Seralini, GM critics and the media to charge that Monsanto used its might to get this paper retracted.”
But that is precisely what happened. In 2013 Hayes officially retracted the Séralini article, later telling the New York Times “Monsanto played no role whatsoever in the decision that was made to retract.” Nonetheless, in his 2013 annual performance summary, Monsanto Toxicology Manager David Saltmiras listed among his accomplishments: “Successfully facilitate numerous third party expert letters to the editor which were subsequently published, reflecting the numerous significant deficiencies, poor study design, biased reporting and selective statistics employed by Seralini.” The website Retraction Watch noted that “An FCT investigation found no evidence of fraud, misconduct, or gross error [in the Seralini paper], which are required by Committee on Publication Ethics (COPE) guidelines for retraction; however, FCT cited COPE guidelines in their retraction notice anyway.”
With this history as background, Monsanto quickly began to marshal its resources in 2014 to fight what they anticipated to be a harmful conclusion of the IARC panel. In an internal October 2014 e-mail, William Heydens wrote “while we have vulnerability in the area of epidemiology, we also have potential vulnerabilities in the other areas that IARC will consider, namely, exposure, genetox, and mode of action…” By February 2015 the company circulated a “Preparedness and Engagement Plan for IARC Carcinogen Rating of Glyphosate.” The plan focused on orchestrating outcry with the anticipated IARC decision, by using “sympathetic scientists, front groups and trade associations to shape public opinion and pressure regulators to ‘remain focused on the science, not the politically charged decision by IARC.’”
Implementation of the plan began immediately. For example, Monsanto’s Eric Sachs suggested to Forbes contributor Henry Miller that he publish an article on the Forbes website refuting the IARC finding even before it would be announced: “Ideally, your article would precede the IARC decision. Why not set the table with the weight of scientific evidence before IARC convenes? Then, regardless of what they do, your article will set the state for a science-based response.” Miller, a Fellow at Stanford University’s Hoover Institute, agreed but requested “a high quality draft” from Monsanto. He then posted that ghost-written draft, with few edits, on the Forbes website on March 17, 2015, under the title “March Madness from the United Nations.” When the ghostwriting was eventually revealed via the discovery documents, Forbes fired Miller and removed all of his posts from the Forbes.com site. But that would hardly be the end of Monsanto’s ghost-writing allegedly “independent” scientific articles.
Another paper appeared in the industry-friendly journal Critical Reviews in Toxicology in March 2015, with co-authors Helmut Greim, David Saltmiras (Monsanto Toxicology Manager) and others. Greim has long been one of Germany’s forefront toxic product defenders. For example, he chaired the Research Advisory Council for the European carmakers’ front organization EUGT during the bizarre “Monkeygate” scandal described in Part I of this post, and worked to defend the unjustified and inconclusive Volkswagen diesel exhaust experiment. The Greim paper claimed to be an independent review of previous rodent studies regarding the carcinogenic potential of glyphosate. Its conclusion: “After almost forty years of commercial use, and multiple regulatory approvals including toxicology evaluations, literature reviews, and numerous human health risk assessments, the clear and consistent conclusions are that glyphosate is of low toxicological concern, and no concerns exist with respect to glyphosate use and cancer in humans.”
But the claim to independence was later destroyed by another William Heydens e-mail from February 2015 revealed in discovery: “A less expensive/more palatable approach [to a helpful paper] might be to involve experts only for the areas of contention, epidemiology and possible MOA [Mode of Action] (depending on what comes out of the IARC meeting), and we ghost-write the exposure tox & genetox sections. An option would be to add Greim and Kier or Kirkland to have their names on the publication, but we would be keeping the cost down by us doing the writing and they would just edit & sign their names so to speak. Recall that is how we handled Williams Kroes & Munro, 2000.” And Saltmiras, ever proud to tout his accomplishments, claimed in an internal August 2015 summary of his work: “ghost-wrote cancer review paper Greim, et al. (2015).”
Also in March 2015 IARC announced that they had determined glyphosate to be “a probable human carcinogen.” Their conclusions were based on “sufficient evidence of carcinogenicity” in studies of lab animals, “limited evidence” in humans, and evidence that glyphosate “caused DNA and chromosomal damage in human cells.” They cited specifically an association revealed in numerous studies between high levels of glyphosate exposure and non-Hodgkin lymphoma (NHL). This link was interesting because the incidence of NHL had been increasing over the preceding few decades, especially among farmers and agricultural workers.
The IARC labeling did not carry with it any ban or regulations on glyphosate usage, but it stimulated roughly 1000 cancer claims to be made in so-called multi-district lawsuits filed against Monsanto and Roundup between 2015 and early 2017, with many of the cases being centralized in a federal court in San Francisco. It is the discovery for these lawsuits that led to the release of the reams of damning internal Monsanto communications over the decades. Monsanto attorneys tried unsuccessfully to keep the documents under seal, arguing that “its internal communications taken individually do not accurately reflect the company’s actions or intentions.” They sure do paint a troubling pattern, though.
By April 2019 the number of lawsuits filed in U.S. courts had grown to 13,000, most on behalf of plaintiffs who had high levels of work exposure to Roundup, and who claimed that Monsanto had been well aware of the toxic nature of Roundup for years, while advertising it falsely. Most of the lawsuits claim that it is not glyphosate alone, but its particular formulation within Roundup – including the tallow amine known as POEA to help glyphosate adhere to plants – that increases carcinogenicity. This same view, by the way, was reflected in another William Heydens internal Monsanto e-mail, this one from 2002: “Glyphosate is OK, but the formulated product causes the damage.”
Monsanto kept up the onslaught. After the IARC conclusion was announced, Monsanto assembled a supposedly independent “expert panel review” to dispute the IARC classification. The panel comprised 16 sympathetic experts, some chosen as friendly private consultants who had done extensive work for Monsanto over the years. Their review was published again in Critical Reviews in Toxicology, in a supplemental volume, claiming that “the data do not support IARC’s conclusion” and that “glyphosate is unlikely to pose a carcinogenic risk to humans.” The authors claimed no conflict of interest, but the discovery documents revealed after the fact that Monsanto had paid for their participation through the consulting firm Intertek and had been “deeply involved in organizing, reviewing, and editing drafts.” When these connections were revealed, even the industry-supported journal that had published the paper demanded an extensive correction to point out the many ways in which the paper’s authors were not at all independent from Monsanto. Monsanto further used the product defense firms Ramboll Environ and Exponent to author additional papers and comments on U.S. regulatory determinations about glyphosate’s carcinogenicity. They sought third-party experts to “blog, op/ed, tweet and/or link, repost, retweet, etc.” in support of Roundup’s safety.
But Monsanto was not content simply to carpet-bomb the scientific literature and social media. They had connections in federal agencies and Congress to exploit in trying to overturn the IARC classification. In yet another 2015 internal e-mail, Monsanto’s chief regulatory liaison Dan Jenkins said that senior EPA official Jess Rowland “could be useful as we move forward with ongoing glyphosate defense.” Rowland headed an EPA Cancer Assessment Review Committee (CARC) that backed the safety of glyphosate in 2016, leading to the agency’s declaration that glyphosate is “not likely to be carcinogenic to humans.”
n a report issued in 2017, a special advisory panel to EPA said they could not fully support the CARC determination. Despite Monsanto’s successful efforts, via front organization CropLife, to have a panel member who concerned them removed before the meeting, some panel members said the research database suggests “a potential for glyphosate to affect cancer incidence.” They claimed that “many of the arguments put forth” by EPA in support of glyphosate’s safety “are not persuasive.” In May 2017, the EPA’s Inspector General launched “an investigation into reports that an EPA employee may have colluded with Monsanto to conduct a biased review of glyphosate.” None of this, however, was sufficient to change the EPA classification, leaving us with the conflict of views summarized in Fig. 9.

While the EPA was considering its Monsanto-friendly classification, a different U.S. health organization, the Agency for Toxic Substances and Disease Registry (ATSDR) within the Department of Health and Human Services, was independently researching the extensive literature to determine glyphosate’s toxicity. Both Monsanto and the EPA were quite concerned that the ATSDR evaluation might be too independent, with Rowland writing to Jenkins in 2015: “If I can kill this I should get a medal.” Despite the best efforts of EPA’s Pesticides Office and current official Mary Manibusan, they were unable to prevent the 2019 release of the ATSDR draft report, whose findings supported the IARC labeling of glyphosate.
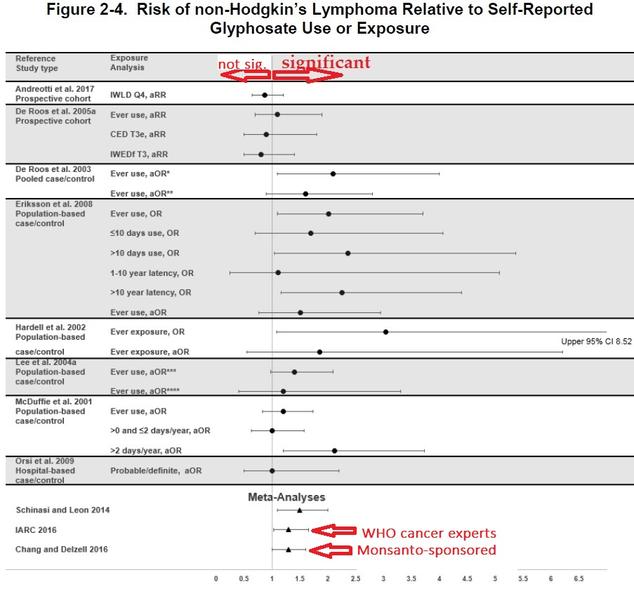
As summarized in Fig. 10, adapted from that report, the weight of the evidence from a variety of studies suggests that glyphosate usage causes a 30-40% increase in NHL incidence, although the existing uncertainties are still large enough that the debate is unlikely to be settled soon. (The glyphosate studies have not had the benefit of anything close to the 69,000-person cohort Rob Bilott provided for PFOA.) In particular, we note that the 2016 meta-analysis carried out by Exponent scientists supported by Monsanto agrees quite well with the meta-analysis done by the IARC panel, but the two analyses still came to opposite conclusions regarding the probability of carcinogenicity. As of now, the ATSDR analysis has not caused EPA to alter its classification.
Monsanto and its now parent company Bayer still faced many lawsuits, and were thus not satisfied with simply avoiding U.S. regulations. They have worked actively to discredit IARC and to push for a reversal of the IARC classification. In 2017 the American Chemistry Council trade organization launched a new front group, the Campaign for Accuracy in Public Health Research for the express purpose of seeking “reform of the International Agency for Research on Cancer’s (IARC) Monographs Program, which evaluates the carcinogenic hazard of substances and behaviors.”
In 2017-18, at the chemical industry’s behest, the Republican leadership of the U.S. House Committee on Science, Space and Technology wrote “several threatening letters to the newly installed IARC director, Elisabete Weiderpass, demanding the agency send a representative to answer questions about the glyphosate decision and the monograph process. These letters raised the possibility of cutting U.S. government funding for the agency and were followed by a legislative effort to actually do so.” They then scheduled a February 2018 hearing entitled “In Defense of Scientific Integrity: Examining the IARC Monograph Programme and Glyphosate Review.” In preparation for that hearing, the minority members of that Committee put out the report “Spinning Science and Silencing Scientists,” which summarized many of the embarrassing Monsanto transgressions revealed in the discovery documents, some of which we’ve discussed here.
When the Republicans lost control of the U.S. House in the 2018 elections, these efforts to overturn the IARC classification ceased. Another casualty was legislation to satisfy various industry lobbying organizations by drastically restricting the rules under which class action and multi-district lawsuits — such thorns in the reputations of DuPont and Monsanto – could be filed. Such a bill, The Fairness in Class Action Litigation Act, was in fact introduced by Republican Bob Goodlatte of Virginia in 2017. The bill passed the U.S. House of Representatives quickly, but was never even brought up for a vote in the Senate Judiciary Committee, let alone the full Senate.
The first three Roundup lawsuits were decided by juries in 2018 and 2019, all going against Monsanto and Bayer. The juries recommended damage awards to the plaintiffs of $289M, $80M and $2B (that’s Billion with a B), though those damages were subsequently reduced by judges to $78M, $26M and $86.7M, respectively. In July 2019, U.S. District Judge Vince Chhabria, who oversaw the cases brought in San Francisco, said that a punitive award was justified because evidence presented at the trial “easily supported a conclusion that Monsanto was more concerned with tamping down safety inquiries and manipulating public opinion than it was with ensuring its product is safe.” The same claim can be made against many toxic product corporations, based on the evidence.
Noting the trend in these settlements, Bayer has agreed, as of June 24, 2020, to settle all the Roundup cases for a sum of $10.9 billion. Settlements at that level will begin to get the attention of toxic product manufacturers, and perhaps will alter their perceptions of the incentives in alerting government agencies to early indications of toxicity. On the other hand, Bayer would have been well aware of the potential liabilities they were taking on when they acquired Monsanto in 2018, and still they judged the acquisition to make a positive contribution to their long-term profits.
As of this writing, the use of glyphosate has been forbidden in Austria and is being phased out in Germany, leading up to a total ban by 2023. Several other countries have either banned or suspended sales or imports of glyphosate. But Roundup and Roundup Ready crops remain highly popular in the U.S. and the EPA is unlikely to reconsider its designation of glyphosate safety any time soon. As in the case of the PFAS chemicals, this example again illustrates that litigation is currently much more effective than U.S. government regulatory agencies in revealing toxicity and punishing bad behavior of the producers.
III. Summary
In the early 1960s there was a U.S. television series named The Naked City, a police procedural about cases in New York City. Every episode ended with the same tag line: “There are eight million stories in the Naked City. This has been one of them.” In the same spirit, there are hundreds of stories about toxic product corporations fighting against epidemiological science, and we have presented two of them. But they serve to illustrate both the corporate mentality and the resources at their disposal to cover up, deny, cast doubt on, and mold public perceptions and outrage about the evidence supporting their product’s health hazards. The profit incentive has led one big industry after another – tobacco, sugar, chemical, pharmaceutical, auto, agribusiness, fossil fuel, etc. – to demand the right to kill us softly by marketing products that have public appeal, but hidden long-term dangers. The industries claim all the while that we in the U.S. have an over-regulated economy, and they have succeeded in co-opting many regulatory agencies and many politicians to adopt this view. The case studies above should hopefully convey the idea that, from a public health perspective, over-regulation is hardly the problem.
Since all these industries follow the same essential playbook, a product defense industry has sprung up to profit from the corporations’ needs. The scientists who work within this product defense industry, using techniques outlined in Part I of this post, have steady and financially rewarding employment, but it comes at the expense not only of consumer health and environmental sustainability. In addition, their work produces cynicism in the general public about the scientific enterprise. Who are people to believe when scientists on both sides of these toxic product battles claim the mantle of “sound science?” Why should the general public then take seriously valid scientific warnings about impending crises such as the COVID-19 pandemic or global warming? Our goal on this site is to expose the elements of the science denial playbook – even when it is exercised by professional scientists who fail to divulge their conflicts of interest – in order to allay some of that self-defeating cynicism.
References:
David Michaels, The Triumph of Doubt: Dark Money and the Science of Deception (Oxford University Press, 2020), https://www.amazon.com/Triumph-Doubt-Money-Science-Deception/dp/0190922664
N. Rich, The Lawyer Who Became DuPont’s Worst Nightmare, New York Times Magazine, Jan. 6, 2016, https://www.nytimes.com/2016/01/10/magazine/the-lawyer-who-became-duponts-worst-nightmare.html
C. Gillam, Whitewash: The Story of a Weed Killer, Cancer, and the Corruption of Science (Island Press, 2017), https://careygillam.com/book
Spinning Science and Silencing Scientists: A Case Study in How the Chemical Industry Attempts to Influence Science, Minority Staff Report of the U.S. House of Representatives Committee on Science, Space and Technology, February 2018, https://science.house.gov/imo/media/doc/02.06.18%20-%20Spinning%20Science%20and%20Silencing%20Scientists_0.pdf?1
David Heath, Meet the ‘Rented White Coats’ Who Defend Toxic Chemicals, https://publicintegrity.org/environment/meet-the-rented-white-coats-who-defend-toxic-chemicals/
https://www.epa.gov/pfas/basic-information-pfas
https://en.wikipedia.org/wiki/Dark_Waters_(2019_film)
https://en.wikipedia.org/wiki/Perfluorooctanoic_acid
https://en.wikipedia.org/wiki/Perfluorooctanesulfonic_acid
https://en.wikipedia.org/wiki/Robert_Bilott
https://www.epa.gov/laws-regulations/summary-toxic-substances-control-act
https://www.tera.org/ART/index.html
https://en.wikipedia.org/wiki/Michael_Dourson
S. Lerner, Trump’s EPA Chemical Safety Nominee was in the “Business of Blessing” Pollution, The Intercept, July 21, 2017, https://theintercept.com/2017/07/21/trumps-epa-chemical-safety-nominee-was-in-the-business-of-blessing-pollution/
https://www.cardnochemrisk.com/index.php?option=com_content&view=article&id=54&Itemid=5
D.J. Paustenbach, J.M. Panko, P.K. Scott and K.M. Unice, A Methodology for Estimating Human Exposure to Perfluorooctanoic Acid (PFOA): A Retrospective Exposure Assessment of a Community (1951-2003), Journal of Toxicological and Environmental Health A70, 28 (2007), https://pubmed.ncbi.nlm.nih.gov/17162497/
https://weinberggroup.com/regulatory-strategy-submissions/
https://cdn.toxicdocs.org/QX/QXnogko6Eaqd8wNkE3Mj7rvRo/QXnogko6Eaqd8wNkE3Mj7rvRo.pdf
Teflon and Human Health: Do the Charges Stick?, https://www.acsh.org/news/2005/03/18/teflon-and-human-health-do-the-charges-stick
http://www.c8sciencepanel.org/prob_link.html
J. Mordock, DuPont, Chemours to Pay $670 Million Over PFOA Suits, The News Journal, Feb. 13, 2017, https://www.delawareonline.com/story/news/2017/02/13/dupont-and-chemours-pay-670m-settle-pfoa-litigation/97842870/
P. Grandjean, et al., Serum Vaccine Antibody Concentrations in Children Exposed to Perfluorinated Compounds, Journal of the American Medical Association 307, 391 (2012), https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4402650/
S. Lerner, Bad Chemistry, https://theintercept.com/collections/bad-chemistry/
https://cdn.toxicdocs.org/OE/OEw5RB2dwanx2vkdJOozxJK2L/OEw5RB2dwanx2vkdJOozxJK2L.pdf
https://gradientcorp.com/services-overview.html
B.D. Beck, Expert Report in the Matter of State of Minnesota vs. 3M Company, https://cdn.toxicdocs.org/Rp/RpyyeQY3o2XjBzbOaKvaX8e0a/RpyyeQY3o2XjBzbOaKvaX8e0a.pdf
E.T. Chang, et al., A Critical Review of Perfluorooctanoate and Perfluorooctanesulfonate Exposure and Immunological Health Conditions in Humans, Critical Reviews in Toxicology 46, 279 (2016), https://pubmed.ncbi.nlm.nih.gov/26761418/
S. Lerner, Lawsuit Reveals How Paid Expert Helped 3M “Command the Science” on Dangerous Chemicals, https://theintercept.com/2018/02/23/3m-lawsuit-pfcs-pollution/
A. de la Garza, Dark Waters Tells the True Story of the Lawyer Who Took DuPont to Court and Won. But Rob Bilott’s Fight is Far From Over, Time Magazine, Nov. 25, 2019, https://time.com/5737451/dark-waters-true-story-rob-bilott/
ATSDR’s Minimal Risk Levels (MRLs) and Environmental Media Evaluation Guides (EMEGs) for PFAS, November 2018, https://www.atsdr.cdc.gov/pfas/resources/mrl-pfas.html?CDC_AA_refVal=https%3A%2F%2Fwww.atsdr.cdc.gov%2Fpfas%2Fmrl_pfas.html
M. Halpern, What Is the Responsible Science Policy Coalition?, https://blog.ucsusa.org/michael-halpern/what-is-the-responsible-science-policy-coalition-here-are-some-clues#:~:text=According%20to%20the%20PowerPoint%20presentation,about%20the%20dangers%20of%20PFAS.&text=detailing%20what%203M%20knew%20about%20the%20chemicals’%20harms.
A. Lustgarten, How the EPA and the Pentagon Downplayed a Growing Toxic Threat, ProPublica, July 9, 2018, https://www.oregonlive.com/today/2018/07/pentagon_epa_downplay_threat_o.html
EPA’s Per- and Polyfluoroalkyl Substances (PFAS) Action Plan, February 2019, https://www.epa.gov/sites/production/files/2019-02/documents/pfas_action_plan_021319_508compliant_1.pdf
EPA Announcement of Preliminary Regulatory Determinations for Contaminants on the Fourth Drinking Water Contaminant Candidate List, https://s3.amazonaws.com/public-inspection.federalregister.gov/2020-04145.pdf?utm_campaign=pi+subscription+mailing+list&utm_source=federalregister.gov&utm_medium=email
https://www.americanchemistry.com/About/
https://www.regulations.gov/document?D=EPA-HQ-OW-2019-0583-0240
https://en.wikipedia.org/wiki/Glyphosate
https://en.wikipedia.org/wiki/Roundup_(herbicide)
D. Main, Glyphosate Now the Most-Used Agricultural Chemical Ever, Newsweek, Feb. 2, 2016, https://www.newsweek.com/glyphosate-now-most-used-agricultural-chemical-ever-422419
U.S. Department of Agriculture, Recent Trends in GE Adoption, https://www.ers.usda.gov/data-products/adoption-of-genetically-engineered-crops-in-the-us/recent-trends-in-ge-adoption.aspx
S.B. Powles, Gene amplification delivers glyphosate-resistant weed evolution, Proceedings of the National Academy of Sciences 107, 955 (2010), https://www.pnas.org/content/107/3/955
https://water.usgs.gov/nawqa/pnsp/usage/maps/show_map.php?year=2017&map=GLYPHOSATE&hilo=H
A. Pollack, Weed Killer, Long Cleared, Is Doubted, New York Times, March 27, 2015, https://www.nytimes.com/2015/03/28/business/energy-environment/decades-after-monsantos-roundup-gets-an-all-clear-a-cancer-agency-raises-concerns.html
https://academicsreview.org/about-academic-review/purpose/
G-E. Séralini, et al., Long term toxicity of a Roundup herbicide and a Roundup-tolerant genetically modified maize, Food and Chemical Toxicology 50, 4221 (2012), https://www.sciencedirect.com/science/article/pii/S0278691512005637
D. Hakim, Monsanto Emails Raise Issue of Influencing Research on Roundup Weed Killer, New York Times, Aug. 1, 2017, https://www.nytimes.com/2017/08/01/business/monsantos-sway-over-research-is-seen-in-disclosed-emails.html
H. Greim, D. Saltmiras, V. Mostert and C. Strupp, Evaluation of Carcinogenic Potential of the Herbicide Glyphosate, Drawing on Tumor Incidence Data From Fourteen chronic/carcinogenicity Rodent Studies, Critical Reviews in Toxicology 45, 185 (2015), https://pubmed.ncbi.nlm.nih.gov/25716480/
J. Ewing, 10 Monkeys and a Beetle: Inside VW’s Campaign for ‘Clean Diesel’, New York Times, Jan. 25, 2018, https://www.nytimes.com/2018/01/25/world/europe/volkswagen-diesel-emissions-monkeys.html
Some Organophosphate Insecticides and Herbicides, IARC Monographs on the Evaluation of Carcinogenic Risks to Humans, Volume 112, https://publications.iarc.fr/549
L. Schinasi and M.E. Leon, Non-Hodgkin Lymphoma and Occupational Exposure to Agricultural Pesticide Chemical Groups and Active Ingredients: A Systematic Review and Meta-Analysis, International Journal of Environmental Research and Public Health 11, 4449 (2014), https://pubmed.ncbi.nlm.nih.gov/24762670/
https://www.cancer.org/cancer/non-hodgkin-lymphoma/about/what-is-non-hodgkin-lymphoma.html
G.M. Williams, et al., A Review of the Carcinogenic Potential of Glyphosate by Four Independent Expert Panels and Comparison to the IARC Assessment, Critical Reviews in Toxicology 46(Sup. 1), 3 (2016), https://pubmed.ncbi.nlm.nih.gov/27677666/
Expression of Concern about ‘An Independent Review of the Carcinogenic Potential of Glyphosate’, Editor-in-Chief and Publisher, Critical Reviews in Toxicology 48, 891 (2018), https://www.tandfonline.com/doi/full/10.1080/10408444.2018.1522786
E.T. Chang and E. Delzell, Systematic Review and Meta-Analysis of Glyphosate Exposure and Risk of Lymphohematopoietic Cancers, Journal of Environmental Science and Health B51, 402 (2016), https://pubmed.ncbi.nlm.nih.gov/27015139/
https://oehha.ca.gov/media/downloads/crnr/glyphosatensrlfsor041018.pdf
C. Gillam, Monsanto Weed Killer Deserves Deeper Scrutiny as Scientific Manipulation Revealed, Huffington Post, March 17, 2017, https://www.huffpost.com/entry/monsanto-weed-killer-deserves-deeper-scrutiny-as-scientific_b_58cc5541e4b0e0d348b34348
FIFRA Scientific Advisory Panel, A Set of Scientific Issues Being Considered by the Environmental Protection Agency Regarding: EPA’s Evaluation of the Carcinogenic Potential of Glyphosate, EPA Report No. 2017-01, https://drive.google.com/file/d/0B-pJR4cGo9ckeXFVVkxpNUwzOEU/view
Toxicological Profile for Glyphosate, ASTDR Draft Report, April 2019, https://www.atsdr.cdc.gov/toxprofiles/tp214.pdf
J. Sass, ATSDR Report Confirms Glyphosate Cancer Risks, https://www.nrdc.org/experts/jennifer-sass/atsdr-report-confirms-glyphosate-cancer-risks
H. Yan, The EPA says glyphosate, the main ingredient in Roundup, doesn’t cause cancer. Others aren’t so sure, CNN, May 3, 2019, https://edition.cnn.com/2019/05/01/health/epa-says-glyphosate-is-safe/index.html
A. Frankel, Class Action Reform Isn’t Dead. It’s Just Not Coming from Congress, Reuters, Dec. 28, 2018, https://www.reuters.com/article/legal-us-otc-classaction/class-action-reform-isnt-dead-its-just-not-coming-from-congress-idUSKCN1OR1G1
Judge Reduces $80M Award In Roundup Case; Cancer Patient, Monsanto Both Consider Appeal, CBS SF Bay Area, July 15, 2019, https://sanfrancisco.cbslocal.com/2019/07/15/80-million-award-reduced-monsanto-roundup-cancer-patient/
Bayer Paying Up to $10.9B to Settle Monsanto Weedkiller Case, New York Times, June 24, 2020, https://www.nytimes.com/aponline/2020/06/24/business/ap-eu-bayer-monsanto-settlement.html
N. Kresge and T. Loh, Bayer Warns on Profit Outlook After $63 Billion Monsanto Deal, Bloomberg News, Sept. 5, 2018, https://www.bloomberg.com/news/articles/2018-09-05/bayer-sees-earnings-lower-after-63-billion-monsanto-purchase
